Title: Ionic Crystals of {[Ni(phen)3]2Ge4S10}-xSol, Showing Solid-State Solvatochromism and Rapid Solvent-Induced Recrystallization
Authors: Wen-Qin Mu, Qin-Yu Zhu, Li-Sheng You, Xu Zhang, Wen Luo, Guo-Qing Bian, Jie Dia
Journal: Inorganic Chemistry
Take Home Importance According to Blogger: A solid material which could rapidly change color depending on what solvent was introduced is reported, with potential sensing applications.
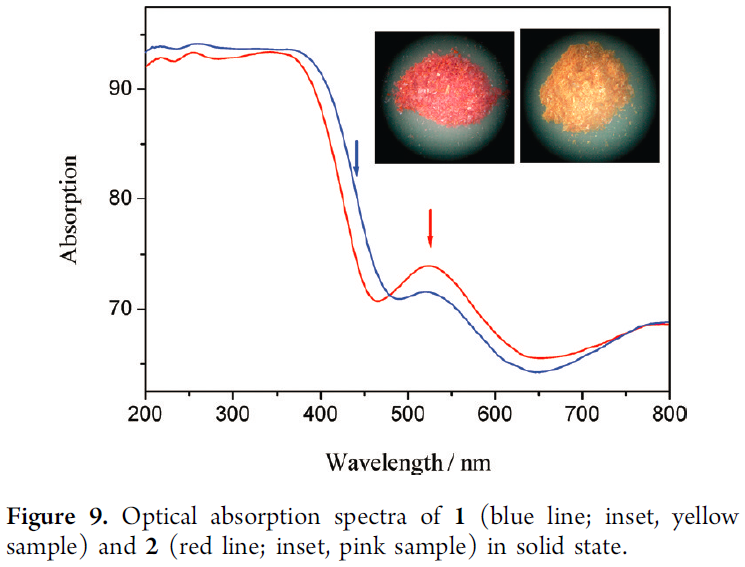
Summary: Materials which rapidly respond to their environment and simultaneously tell you that something is different have applications in sensing. As an example, some fluorescent materials exhibit ‘turn-off’ fluorescence when TNT molecules bind to them, which is good for figuring out if you should be watching your step. Here, the authors report a novel material which exhibits solid state and reversible solvatochromism, or an ability to change color based on the polarity of the solvent present, as shown in Figure 9. The polarity of the solvent may shift up or down the energies of the ground and excited states because the localized charges in solvents (i.e. negative on oxygen in water and positive on the hydrogens in water) interact with the partially charged molecular orbitals of the ground and excited states. The result is a slightly different energy gap between ground and excited state and consequently a different color. Solvatochromism has a simplistic beauty in that you just have to watch it change color to know something has happened.
The materials reported are ionic thiogermanates, meaning a) that it is a two component system with equal amounts of positive and negative components, b) has sulfur in it (the ‘thio’ part), and c) contains germanium. Specifically, the authors prepared crystals of [Ni(phen)3]2Ge4S10-xSolvent (the amount and type of solvent was variable) by a liquid-liquid diffusion method where the two starting materials were dissolved in separate, miscible solvents and carefully layered. Over time the two layers slowly diffuse into each other permitting slow crystallization of the product, and as a rule of thumb slower growth = higher quality crystals. This technique can be visualized in the ‘Liquid-Liquid Diffusion’ section of the MIT X-Ray Crystallography website on growing crystals along with other methods.
What makes this article interesting is that the solvatochromism shown occurs in the solid state instead of in solution phenomenon. Key to this behavior is the large amount of solvent inside the materials. This solvent is held in pores and ‘tunnels’ running throughout the crystals. So, while the ionic thiogermanates remain crystalline, the ability of solvents of varying polarity to travel in and out results in a solution-like environment. Figure 9, reproduced here, nicely summarizes the effect: yellow crystals contain 4 molecules of methanol and 12 molecules of water per unit cell and the pink crystals contain 24 molecules of water per unit cell. Beyond showing that these materials change color when exposed to different solvents, the authors studied how rapidly the color changed based on how bulky the introduced alcohol was – with bulkier alcohols like isopropanol exchange occurs less quickly, indicated that solvents actually need to travel into the pores to induce an effect. Below is a video from the paper demonstrating the solvatochromism behavior.
[youtube http://www.youtube.com/watch?v=USO9FxWJtlM&w=280&h=210]
video supplied in the paper’s supporting information
Figures 9 and Video (ic2014682_si_005) Reprinted with permission from Mu et al., J. Am. Chem. Soc., 2012, DOI: 10.1021/ic2014682. Copyright 2012 American Chemical Society.