Apply to join our writing team (rolling basis)!
Would you trust a bacterium to deliver your medicine?
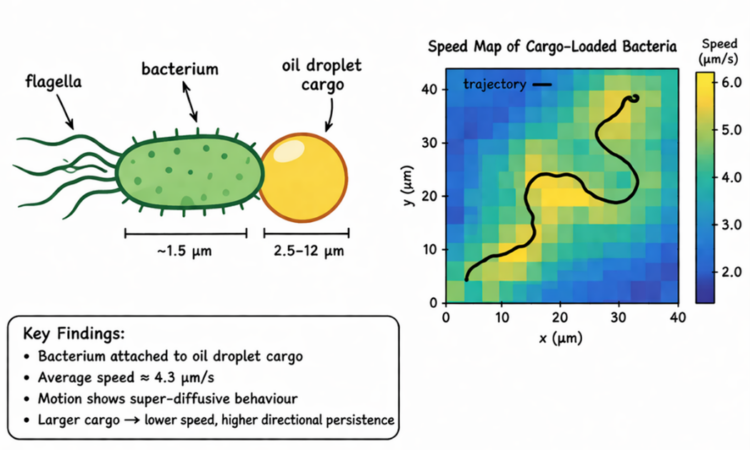
Title: Swimming statistics of cargo-loaded single bacteria Authors: P. Prakash, A. Z. Abdulla, V. Singh, M. Varma Journal: Soft Matter Year: 2020 Featured image adapted from an AI image generator source and Prakash et. al. What if the next breakthrough in drug delivery does not come…
When AI Becomes the Pathologist’s Assistant
What if AI could spot cancer patterns invisible to the human eye? The CHIEF model analyzes tens of thousands of pathology slides to identify tumor types, predict genetic mutations, and estimate patient outcomes, pushing cancer diagnostics into a new era.
Mirroring Nature: L-Cellulose and the Elution Revolution
Have you ever accidentally tried to put a right-handed glove on your left hand? Even though the two gloves look identical at first glance, you can’t put the right-handed glove on your left hand.
Salt of the Earth: Why do some plants grow and others either when the salinity of farm fields increases?
Title: Growth, Photosynthetic Performance, and Water Relations of Weeds under Salt Stress Journal: ACS Agricultural Science and Technology Authors: Jesley Nogueira Bandeira, Lyandra Maria de Oliveira, Larissa Raquel Fagundes Costa Bezerra, Ariana Carvalho Pinto, Ruana Karoline Viana Pereira, Valéria Maria Pedroso de Moraes, María Carolina Ramírez…
How microbes co-break down a dietary molecule
Ergotheinene, a molecule found in mushrooms, is used by multiple gut bacteria.
How Chemists Design New Molecules (and Make Catalysts Work in Water)
Have you ever looked at a complicated molecule and wondered who comes up with it? In the paper by Rolf Breinbauer and colleagues at the Technical University of Graz, the problem was this: how can we attach small chemical tags to proteins without damaging them?
ENGINEERING THE TUMOR MICROENVIRONEMNT
Article: Development of a Hydrogel Platform with GBM and Microglia: A Potential Glioblastoma Tumor Model Authors: Seyma Isik, Deniz Yucel, Vasif Hasirci In: ACS Applied Bio Materials The brain is largely made up of cells that reside in a space called the extra-cellular matrix (ECM). This…
How Cells Handle Damaged Proteins: Two Hidden Members of the Cleanup Crew
Inside every cell, thousands of proteins are made every second. Sometimes a protein folds incorrectly. It can tangle or stick to other misfolded proteins, forming clumps that block normal cell functions. Cells have a cleanup system made of helper proteins.
New Sensor Helps Reveal Secrets of Wound Healing
Title: Application of aptamer – carbon hybrid materials for electrochemical detection of wound healing biomarker – TNF-α protein Authors: Julia Czopinska, Filip Budny, Andrzej Peplowski, Anna Sobiepanek, and Marta Jarczewska. Journal: Bioelectrochemistry Year Published: 2025 Cover Image Credit: Saulhm, wikicommons For many of us, most…
Beyond Particle Mass: Linking Chemical Composition to Air Quality Health Effects
Considering the chemical composition of atmospheric particulate matter pollution could provide valuable health information to inform future policy.
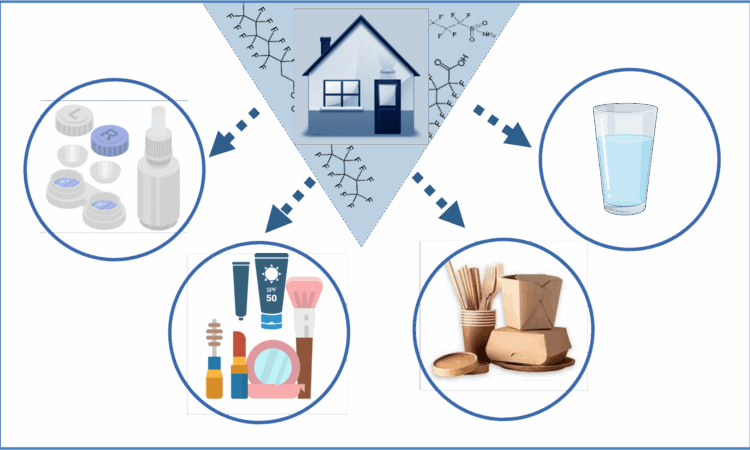
The Hidden Chemistry in Your Routine: Inside Beauty and Hygiene Products
You might expect industrial chemicals to show up in factories or fire fighting gear, but not in your mascara, contact lenses or even your toothpaste! Behind convenience and performance of these personal care products, many of them may contain PFAS. PFAS or per- and polyfluoroalkyl substances…
Tiny Microbes, Big Impact: Can Gut Bacteria Prevent and Treat Obesity?
Title: Human gut microbial aromatic amino acid and related metabolites prevent obesity through intestinal immune control Authors: Zengliang Jiang, Liuqing He, Diyin Li, Laibao Zhuo, Lingjun Chen, Rui-Qi Shi, Jianhua Luo, Yuhui Feng, Yuhui Liang, Danyang Li, Xiao Congmei, Yuanqing Fu, Yu-ming Chen, Ju-Sheng Zheng, Liang…
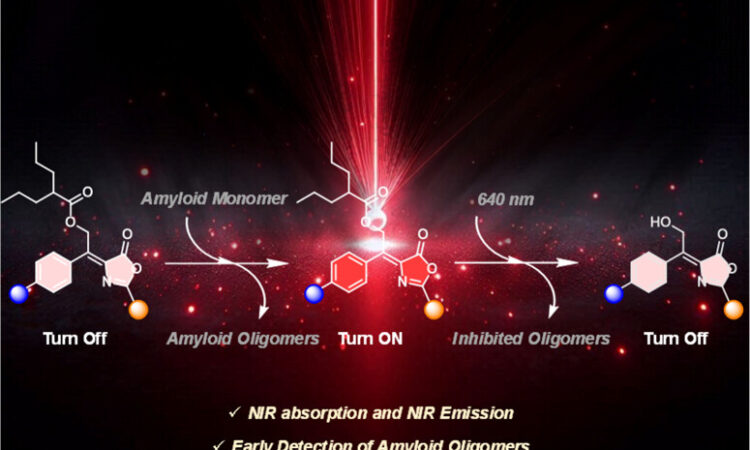
New Light-Activated Molecules Tackle Alzheimer’s Protein Clumps
Inspired by Jellyfish Glow, Scientists Design Light-Sensitive Alzheimer’s Therapy
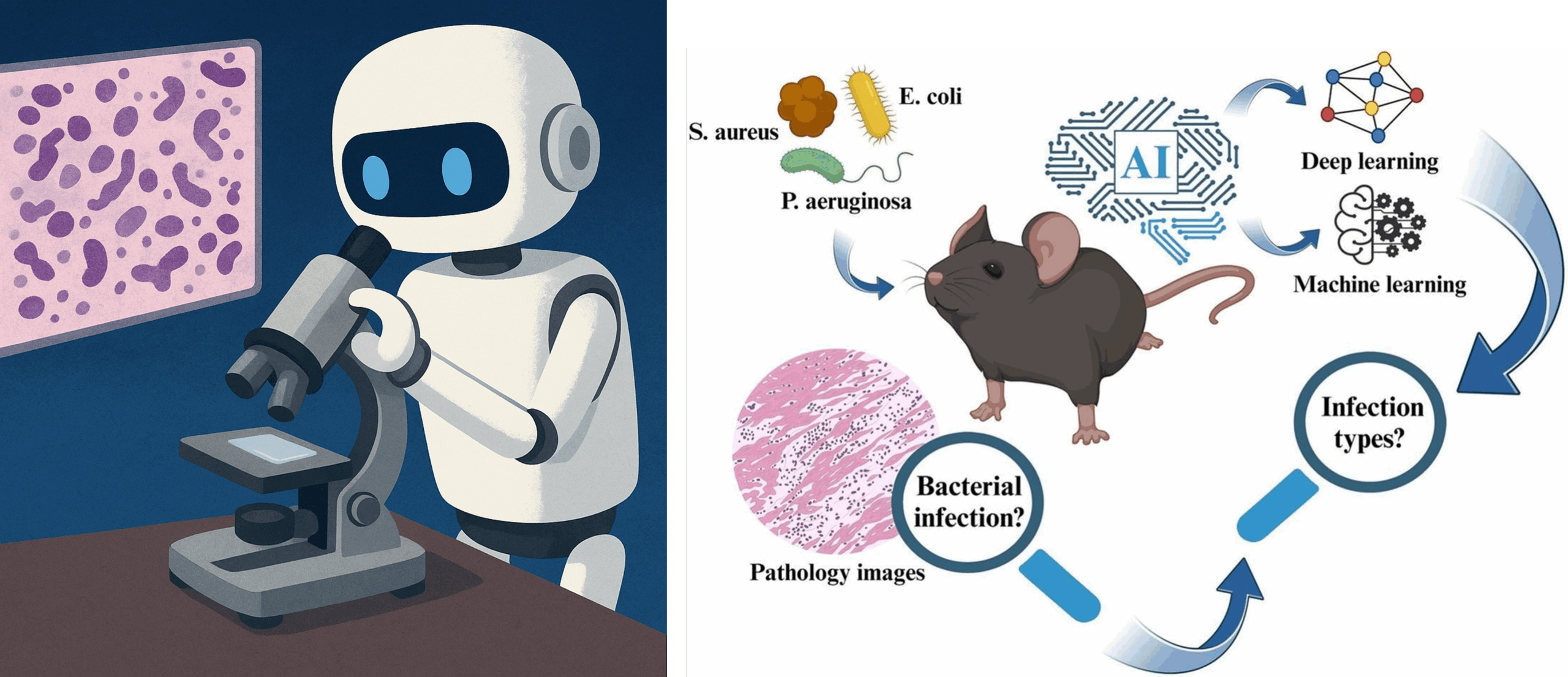
Will AI Become Our New Pathologist?
Title: Using pathology images and artificial intelligence to identify bacterial infections and their types Authors: Xinggong Liang, Gongji Wang, Zhengyang Zhu, Wanqing Zhang, Yuqian Li, Jianliang Luo, Han Wang, Shuo Wu, Run Chen, Mingyan Deng, Hao Wu, Chen Shen, Gengwang Hu, Kai Zhang, Qinru Sun, Zhenyuan…
A simple molecule improves the NMR “fingerprint” of proteins for better quality control of biologics
Scientists expand the current toolbox for good quality control measures of our protein-based treatments by adding a single small molecule to enhance protein structure tracking.
How Exosomes Are Transforming the Fight Against Alzheimer’s Disease
Exosome-Packed Sensors Revolutionize Early Detection of Alzheimer’s and Brain Health Monitoring
Are magnetic nanoparticles the next answer to remove biofilms?
Title: A New Tool to Attack Biofilms: Driving Magnetic Iron-oxide Nanoparticles to Disrupt the Matrix Authors: Jie Li, Rachel Nickel, Jiandong Wu, Francis Lin, Johan van Lierop and Song Liu Journal: Nanoscale Year: 2019 Featured image adapted from an AI image generator source and Li et…
Greenhouse Gasses Bubbling Up From Deep Below Arctic Lakes
This source of greenhouse gasses could act as an important feedback as the arctic warms.
Fishing for Personalized Cancer Treatments
Zebrafish may help clinicians find the most effective chemotherapies for patients.
New Strategy to Clean Fragile Art
Article Title: New sustainable polymers and oligomers for Cultural Heritage conservation† Authors: Damiano Bandelli, Rosangela Mastrangelo, Giovanna Poggi, David Chelazzi and Piero Baglioni Year Published: 2024 Journal: Chemical Science Article DOI: https://doi.org/10.1039/D3SC03909A Cover image credit: RichardMcCoy Paintings, murals, and other pieces of art held in museums…
B is for Battery: Storing Clean Energy with Vitamin B
Article: Exploration of Vitamin B6-Based Redox-Active Pyridinium Salts Towards the Application in Aqueous Organic Flow Batteries0 Authors: Anton A. Nechaev, Gabriel Gonzalez, Prachi Verma, Vsevolod A. Peshkov, Anton Bannykh, Arsalan Hashemi, Jenna Hannonen, Andrea Hamza, Imre Pápai, Kari Laasonen, Pekka Peljo, & Petri M. Pihko Journal:…
Your Coffee break could be a new weapon against Cholera
Title: Potential Antimicrobial Properties of Coffee Beans and Coffee By-Products Against Drug-Resistant Vibrio cholerae Authors: Anchalee Rawangkan, Achiraya Siriphap , Atchariya Yosboonruang , Anong Kiddee , Grissana Pook-In, Surasak Saokaew, Orasa Sutheinkul and Acharaporn Duangjai Journal: Frontiers in Nutrition Year: 2022 Featured image adapted from an…
Coastal Permafrost Erosion: A Climate Feedback
As our planet warms, understanding carbon in the arctic is crucial.
Engineering a Synthetic Cell: Replicating Nature’s Architecture
In their quest to replicate a biological cell, this group of researchers have successfully engineered a system complete with a ‘nuclear’ and a ‘cytoplasmic’ compartment! Moreover, it allows macromolecules to shuttle between the compartments, replicating a key biological functionality.
Keeping It Green While Making It Sweet: More Sustainable Chocolate Made with the Whole Cocoa Fruit
Most commercial dark chocolate combines the fruit from the cocoa nut with sugar from sugar beets. But now, Swiss scientists show how we can use the outer layer of the cocoa pod to sweeten our delectable treat.