| Paper | Host-Selective Phytotoxins Incorporating the Epoxy-Triene-Decarboxylate Moiety Function Through Hijacking of the Plant-Microbe Interaction system |
| Authors | Minoru Ueda, Nobuki Kato, Yoshinori Kurata, Masaki Imai, Gangqiang Yang, Keigo Taniguchi |
| Journal | ACS Chemical Biology |
| Year | 2023 |
| URL | https://pubs.acs.org/doi/10.1021/acschembio.2c00777 |
While we’ve found many applications for fungi, from gastronomical to psychedelic, the mushroom kingdom remains a key threat to our livelihoods—and I don’t just mean to athletes. Fungi can cause devastating blights, such as the Victoria blight that wiped out the US supply of oats in the 1940s or the potato late blight which caused Ireland’s Great Famine in 1845. Part of their deadliness originates from their selectivity—their fungal toxins have a single target and do not infect a wide variety of plants.
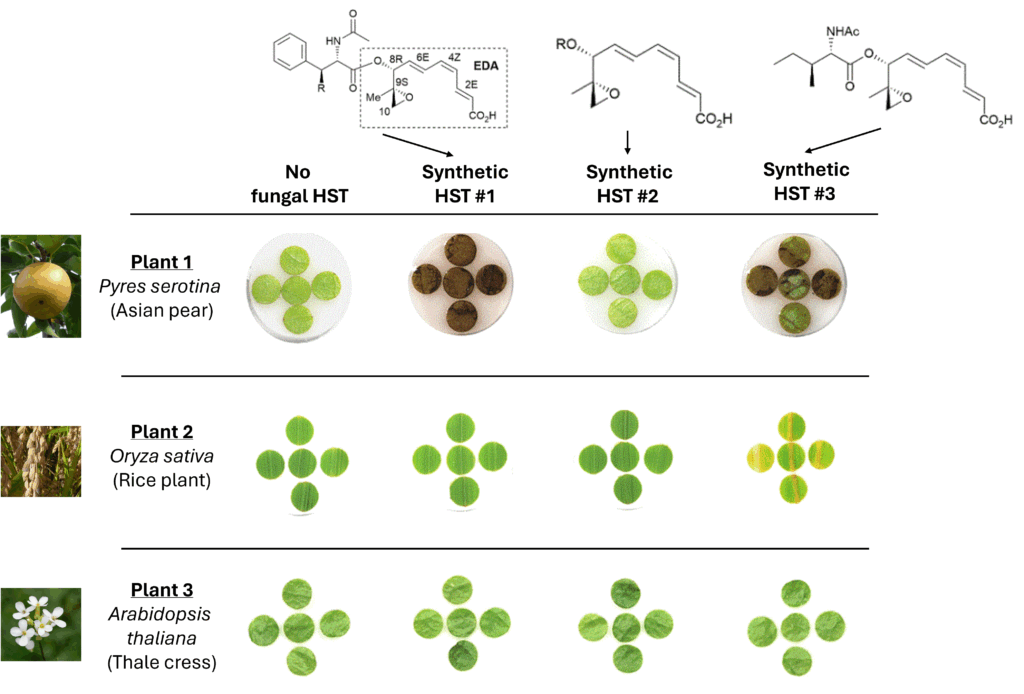
For example, the fungal pathogen Alternaria alternata (abbr. A. alternata) only infects the Japanese pear tree by producing host-selective toxins or HSTs. These chemicals have a specific blueprint that allows the fungi to hijack the pear tree’s immune system and turn it off. However, scientists wondered how this chemical was able to achieve such a thing, as nothing about the chemical itself suggested how it worked. As plants currently used to probe HST functions could not be infected by the highly selective fungi of interest, scientists at Tohoku University in Japan designed a new plant tool—a leaf disc assay.
But first, the scientists had to create a library of different “model” HSTs based on the A. alternata original to test their assays. Prior work demonstrated that the epoxy-triene decarboxylate (EDA) motif of the HST molecule, shown in the dotted box in Fig 1, imparts toxicity towards to the Asian pear. Therefore, they designed 13 HSTs that preserved the EDA motif and varied the acyl-connected section to determine how host selectivity was obtained. The scientists then introduced the panel of HSTs to three plants: the Asian pear, a rice plant, and thale cress. The first was the original fungi target and the other two were model plant organisms, meant to show when the host selectivity changed between the different HSTs.
If the new HSTs worked like the original, only the Asian pear would show signs of death, represented by yellowing or browning leaves, and not impact the rice plant or thale cress. Synthetic HST #1 caused browning in the Asian pear but not the other two – which meant it resembled the native HST and told us no new information. However, the synthetic HST #2, which contains an acetyl glycine functional group, could no longer infect the Asian pear, as shown by the third column. In contrast, by changing the attached functional group to an acetyl isoleucine, HST #3 could infect the Asian pear and the rice plant, expanding its host selection capability.
Following this intriguing result, the scientists analyzed HST #3 further to determine how the HST toxic gain-of-function worked in the rice plant. They found out that this HST, dubbed AcIle-EDA, caused extensive production of reactive oxygen species (ROS) by over-activating an enzyme involved in the plant’s innate immune system. ROS are dangerous as they can cause irreversible damage to DNA and proteins, promoting their premature degradation. Furthermore, the scientists traced the activation of this enzyme back to a specific receptor, called leucine rich repeat or LRR, that is known to interact with small molecules via specific domains, such as those conserved in HSTs. LRRs are also one of the largest receptor families in plants. Their results illustrate how targeting the LRR receptors could be a viable anti-fungal prevention for the Asian pear and highlighted their expanded leaf disc assay as a visual way to study HST structure–activity relationship.

