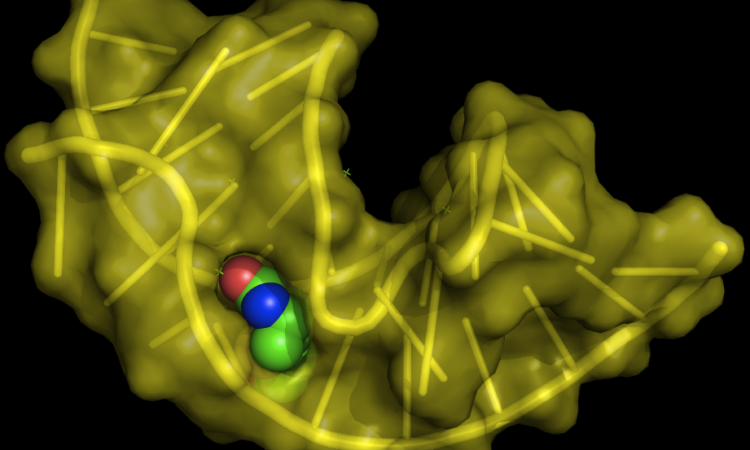
With a growing interest in the field of RNA-targeted therapeutics, robust platforms to study RNA small-molecule interactions are needed. Read about the latest endeavor here!
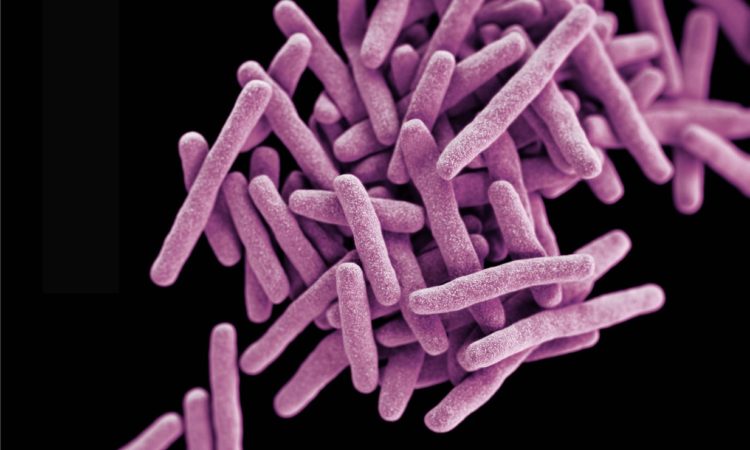
A Closer Look At The Walls of The Toughest Microbes
Mycobacteria are tough, pathogenic microbes that shield themselves with a hardy envelope known as the mycomembrane. Little is still known about the proteins that build or interact with this envelope, but these researchers are up to the challenge.
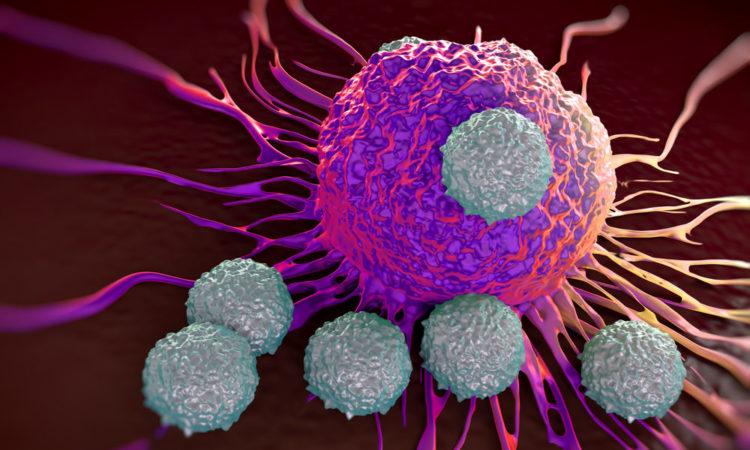
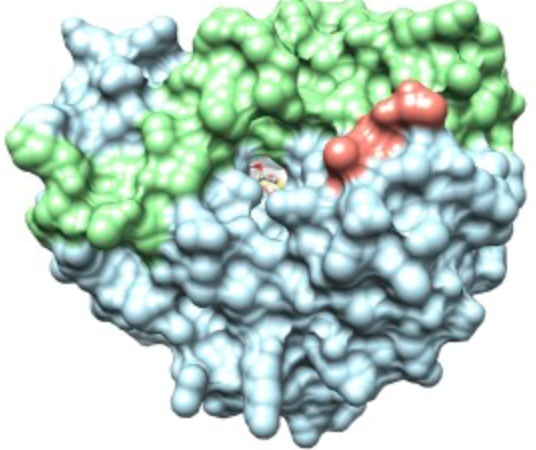
Expanding the Chemical Toolkit to Study Immune Cells
Their highly specialized roles of immune cells also mean they have molecular machineries that are a bit different from those in other cells, Find out here how researchers are using chemistry to advance our knowledge of one of such components, the immunoproteasome.
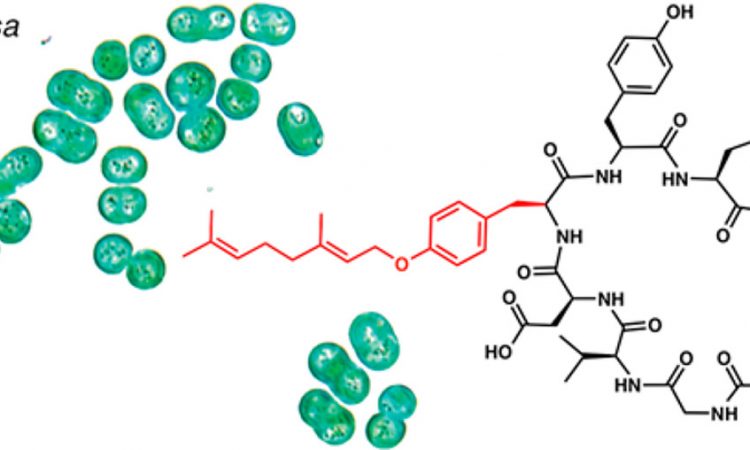
Controversial Mushrooms Can Be Useful After All
With a renewed interest in psilocybin — the psychedelic substance present in magic mushrooms — by the medical community, the Weng group at MIT sets up to study one of the enzymes that makes it.
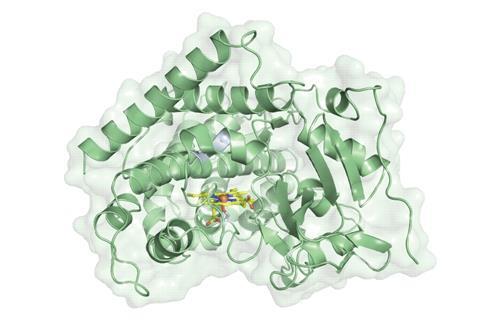
Enzymes Get Closer to Traditional Organic Chemistry – The Case of Reductive Amination
A clever, two-part biocatalytic strategy grants access to products of reductive amination that can be troublesome to obtain through more traditional synthetic methods.
New Insights on The Elusive Protein Sulfinylation
Proteins bear a staggering collection of small chemical modifications that have large effects on their function. This research provides an elegant method to study cysteine sulfinylation, a chemical mark that has proven to be pretty elusive.
Painting a Fuller Picture in Medicinal Chemistry
Title: Synergistic effects of stereochemistry and appendages on the performance diversity of a collection of synthetic compounds Authors: Stu Schreiber et al. Journal: Journal of the American Chemical Society https://pubs.acs.org/doi/10.1021/jacs.8b07319 Year: 2018 The ability to rapidly evaluate what a chemical compound does to a cell, and…
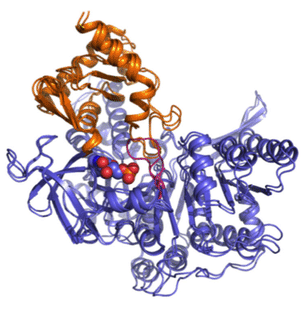
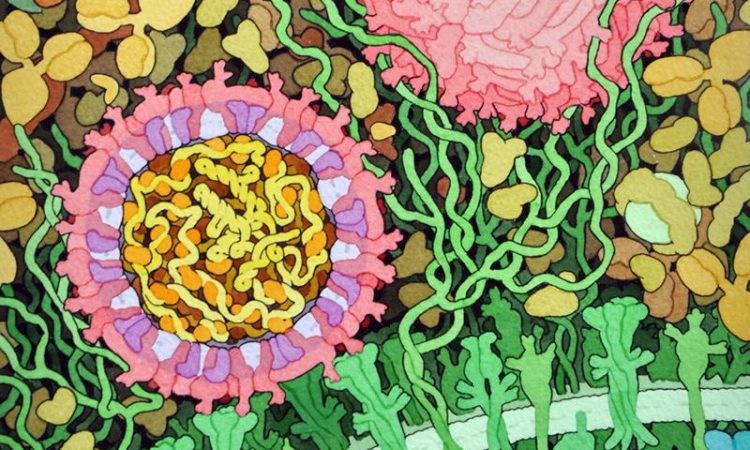
New Findings on Viral Inhibition
Successful resistance to a viral infection requires the host to deploy incredibly intricate biological tactics that somehow selectively inhibit key processes in the viral lifecycle. In this paper, researchers delve deeper into the molecular mechanisms of one of such resistance mechanisms!
Discovering More Structural Diversity in Bacterial Natural Products
Microorganisms are particularly remarkable at churning out structurally challenging small molecules with interesting biological functions. In this work, an unprecedented chemical transformation in one such natural products is discovered and characterized.
Evolving Proteins to Make Tiny Carbon Rings
Learn how researchers at Caltech artificially evolved proteins to synthesize some of the most challenging tiny molecules in organic chemistry!
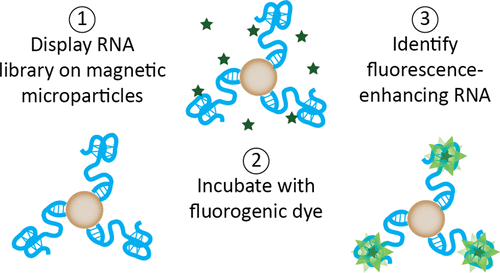
Fluorogenic RNA Molecules Expand the Imaging Toolbox
Nucleic acids are incredibly versatile molecules that can perform functions way beyond their canonical roles in biology. Here, RNA sequences are “evolved” to bind and enhance the fluorescence of a small-molecule dye, welcoming the idea of RNA for robust fluorescence imaging!
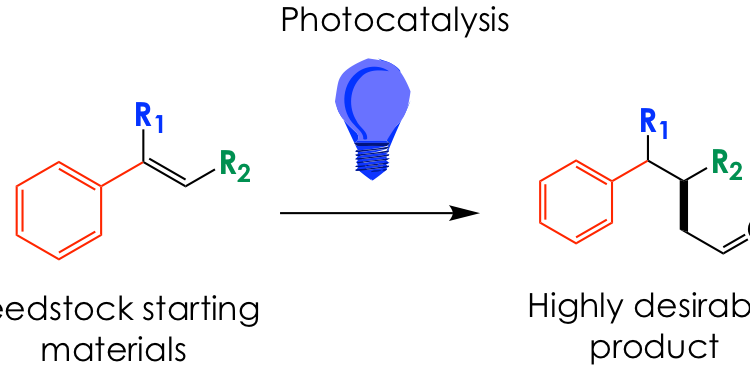
Shining Light on Aldehyde Synthesis
Photoredox catalysis is at it again! This time it is used to synthesize polysubstituted aldehydes – highly useful building blocks – from readily available styrenes and vinyl ethers.
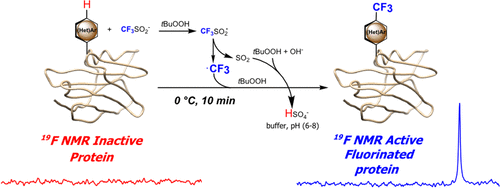
A Radical Way to Label Proteins
Read about this new approach to selectively install fluorines into proteins using a mild radical source.
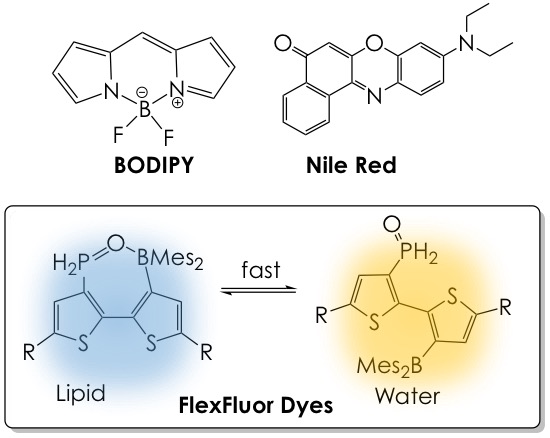
Flexible Fluorescent Dyes for the Detection of Lipids
The functional repertoire of lipids grows to more impressive heights as scientists continue to unravel the substantial functions of these biomacromolecules in cell biology.
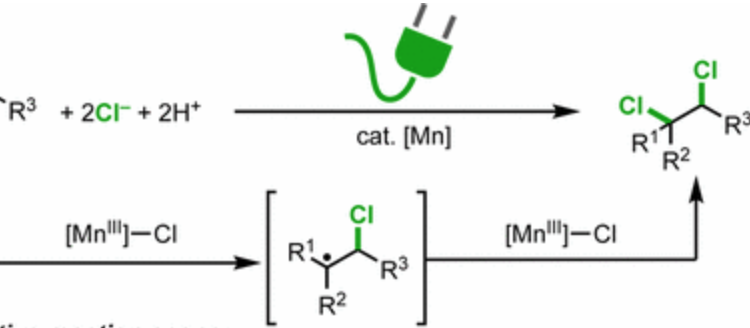
A New Dichlorination of Alkenes: A Radical Twist to a Long-sought Transformation
Title: Electrocatalytic Radical Dichlorination of Alkenes with Nucleophilic Chlorine Sources Authors: Niankai Fu, Gregory S. Sauer, and Song Lin Journal: Journal of the American Chemical Society http://pubs.acs.org/doi/10.1021/jacs.7b09388 Year: 2017 The unsaturated bond of an alkene is one of the most exploited functionalities inside the organic chemist’s…
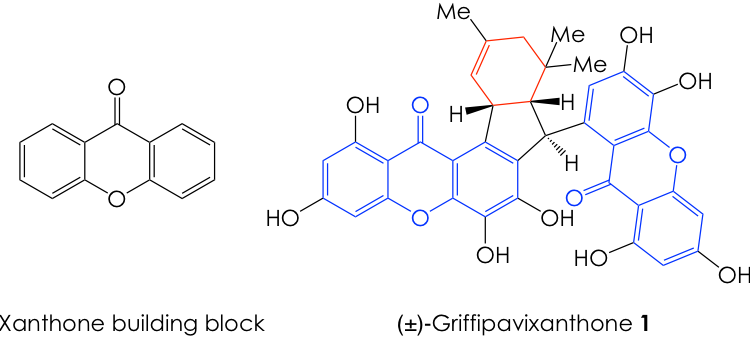
The First Chemical Synthesis of Griffipavixanthone: Bringing out Intellectual and Artistic Virtuosity in Synthetic Chemistry
How do synthetic chemists use fundamental, yet robust reactions to chemicals stitch together smaller fragments large and complex natural products? The Porco’s Lab latest work might give you a taste of what it’s like to exercise the art of total synthesis with a hint of inspiration from biology.