Researchers have developed a new metal-organic framework that can harvest clean water directly from the air, even in arid climates.
Just Breathe: Detecting Diseases with Exhaled Breath
Researchers utilize a mass spectrometry method to detected biomarkers relevant for identifying respiratory diseases in exhaled breath.
Are you inhaling plastics?
Title: Microplastics in the atmosphere of Ahvaz City, IranAuthors: Sajjad Abbasi, Neamatollah Jaafarzadeh, Amir Zahedi, Maryam Ravanbakhsh, Somayeh Abbaszadeh & Andrew Turner.Journal: Journal of Environmental SciencesYear: 2023 Since the invention of plastics, humanity has found countless uses for this class of wonder-materials. From drinking straws to…
Going green: Making a vital chemical from nothing but water and sunlight
Hydrogen peroxide can be produced from water using green electrical energy, offering an environmentally friendly route to a vital material.
Clothing Dye or Light Source? Perhaps Both.
Title: From Textile Coloring to Light-emitting Electrochemical Devices: Upcycling of the Isoviolanthrone Vat DyeAuthors: Tatiana Ghanem, Kwang Keat Leong, Hwandong Jang, Alexis Hardouin, Dr. Philippe Blanchard, Prof. Dominik Lungerich, Dr. Pierre Josse, Prof. Eunkyoung Kim, and Dr. Clément CabanetosJournal: Chemistry, An Asian Journal Featured Image: Textile and Fibre Technology, CSIRO…
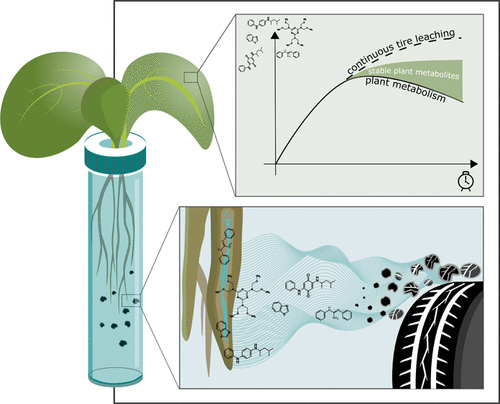
Tires in your salad? Lettuce absorbs pollutants from tire wear, study finds
Researchers show that lettuce plants can metabolize tire wear products into new compounds with unknown toxicities.
Toxic food for thought: detecting illegally added chemicals in fresh food products
Researchers develop a novel method for detecting illegally added formaldehyde in food products.
Jet Engine Oils Cause Ultra-Fine Particle Concentrations to Take Off
Title: Nucleation of jet engine oil vapours is a large source of aviation-related ultrafine particles.Authors: Florian Ungeheuer, Lucía Caudillo, Florian Ditas, Mario Simon, Dominik van Pinxteren, Doğuşhan Kılıç, Diana Rose, Stefan Jacobi, Andreas Kürten, Joachim Curtius & Alexander L. VogelJournal: Communications Earth & EnvironmentYear: 2022 Earth’s…
Unexpected gold nanoparticles arise in ancient buildings through centuries of degradation
New research reveals plausible degradation pathways of metallic gold into nanoparticles through unforeseen corrosion!
Wearable technology powered by your own body heat
Thermoelectric materials can convert heat into electricity, which gives us the possibility of charging smart devices as we wear them. The latest research is working to make these lightweight and flexible so make it cheaper and easier to power wearable electronic devices.
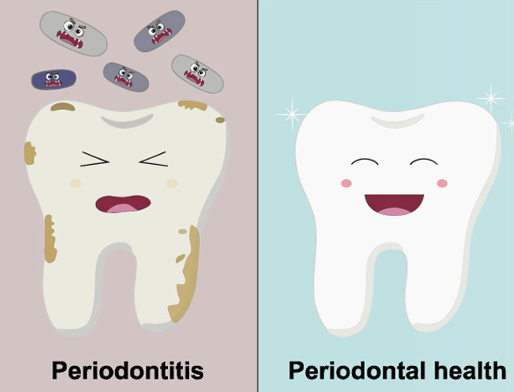
Smile Big!… An Interesting Family of Compounds Can Help Fight Periodontal Diseases
Smile Big! Researchers have developed new compounds that could help fight periodontal diseases.
Electrified nanopipettes decrease diagnostic time for amyloid diseases from days to hours
Researchers at the University of Montpellier develop a diagnostic test for Parkinson’s disease with a glass pipette and electric current that’s quick, accurate and inexpensive.
From harmful emissions to vital materials: how electrochemistry is making the most of carbon dioxide waste
Carbon dioxide can be converted into useful materials using electrochemistry. This is doubly advantageous, since we can stop carbon dioxide entering the atmosphere and also produce something useful that usually comes from fossil fuels.
Polymers efficiently extract water from air in arid conditions
A new and sustainable polymer gel unlocks water capture efficiencies never seen before!
Molecular Docking Techniques Reveal Molecular Interactions Between Novel Antifreeze Peptides and Ice Crystal Growth
The use of molecular docking techniques aid investigation of molecular mechanisms and development of novel antifreeze peptides for applications in the food industry.
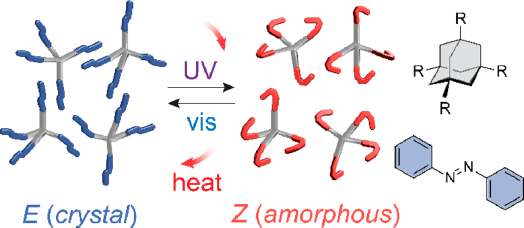
Sunlight to heat: New solid materials store and release solar energy by rotating bonds
A new class of light-absorbing solids isomerizes to store light energy as heat.
A Peek Inside a Fast-Charging Battery
Researchers can now watch what happens to particles inside a battery in real time.
Fungi make molecules for batteries
A active material for redox flow batteries was synthesized from fungi to create a renewable feedstock.
Miniature Device Detects Antibiotic-Resistant Bacteria With Ease
A hand-held device to detect antibiotic resistant bacteria? Some researchers in Singapore made this idea a reality.
A shipwreck gives insight into plastic degradation
Researchers use shipwreck containing plastic pellets as a natural experiment for plastic degradation.
Soaking up the sand: using solvents to detect chemical warfare agents
Researchers explore the best options for extracting nitrogen mustards, a chemical warfare agent, from sand.
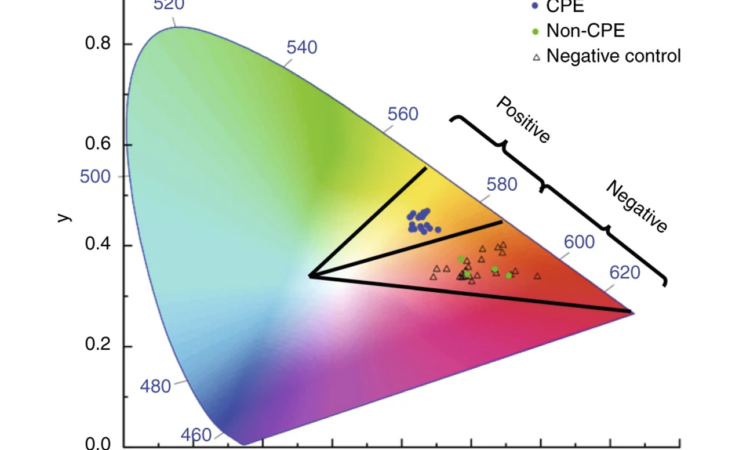
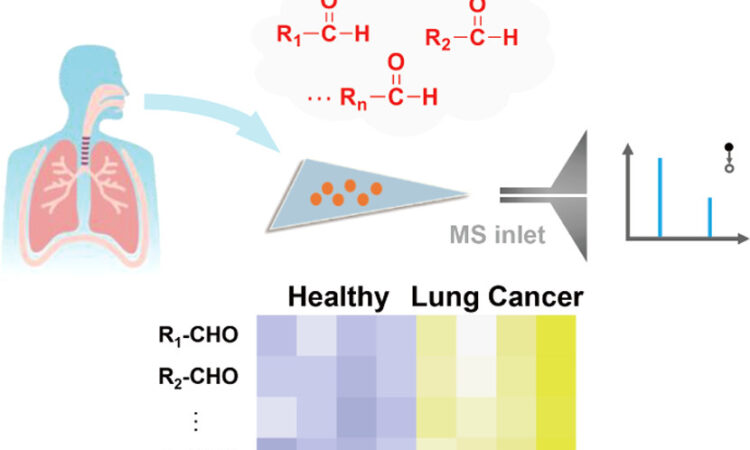
Just Breathe… A Cutting-Edge Method to Diagnose Lung Cancer
Can exhaled breath be used to diagnose lung cancer? A recent study explores this possibility.
Atmospheric chemistry to make you go OOOH!
Recent findings that reveal a previously neglected group of compounds in our atmosphere.
Hydrogen Swap: Exploring the Most Basic Reactions
The most basic reactions are movements of hydrogens — protons — from one molecule to another. Insuati et. al. explored hydrogen tunneling in a dimer of a common organic molecule to look at the very fundamentals of reactivity.
Identifying Receptors Involved in SARS-CoV-2 Infection via Proximity Labeling
SARS-CoV-2 attaches to human cells by binding its spike protein to ACE2 receptors — but could other receptors be involved in this process?