Title: Otherwise Unstable Structures Self-Assemble in the Cavities of Cuboctahedral Coordination Cages
Authors: Felix J. Rizzuto, William J. Ramsay, and Jonathan R. Nitschke
Year: 2018
Journal: Journal of the American Chemical Society
DOI: 10.1021/jacs.8b07494
Over the last millennium, the scientific world has progressed in leaps and bounds, with great discoveries only serving to open more doors, allowing us to explore unanswered questions. Given the monumental discoveries we have seen, one question remains: Will this ever end? At the current technological levels, the simple, straightforward inorganic metal-ligand complexes have been found, and discovery of novel complexes requires some level of ingenuity.
In this work, the Nitschke group at Cambridge University effectively demonstrates a novel approach to the synthesis of complexes by utilizing constraints to aid the formation geometries that would typically be unfavorable. They use a cage-like, octahedral molecular structure, not unlike a cube, which allows for the binding of additional molecules that are not part of the initial structure, also referred to as guest molecules, at its center. The rigidity of the structural skeleton, as well as the limited size of the interior, allows for the formation of specific structures within these bounds.
These new complexes can only be formed within the cage and are unable to exist independently. This is due to the environment-specific interactions induced by these cages, which force the components into a unique formation. The cages act like a “cube trap”, where the cubes enable the formation of new complexes via the restricted geometry and internal surface electronics.
The cubic structure of the cage has 3 sets of parallel walls, which direct guest molecules within the internal cavity. The researchers incorporated different metals in the middle of the facial ligands to tune the electronic interactions with the guests. These variations allow them to increase the stability of the complex by utilizing pre-existing knowledge of chemical and steric interactions.
The researchers demonstrated that it is possible to tune the cage properties, such as shape and symmetry, by incorporating different metals into the cube trap’s structure. Additionally, distinct types of internal assembly were achieved, with the most notable being a threaded ring within the cube, forming what is basically a fidget spinner on a molecular scale.
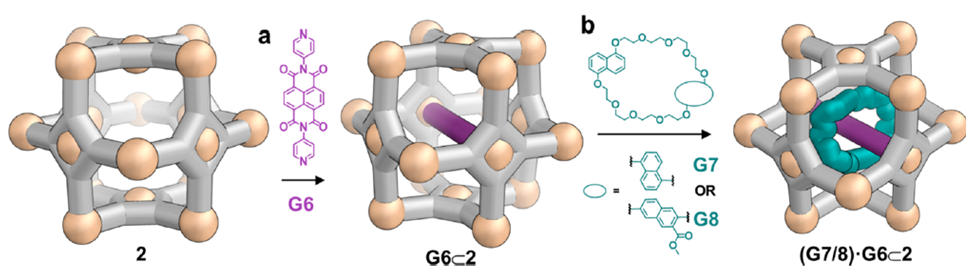
Figure 1. The formation of an encapsulated rotaxane; alternatively – a molecular fidget spinner.
Adapted with permission from J. Am. Chem. Soc., 2018, 140 (36), pp 11502–11509. Copyright 2018 American Chemical Society.
In this experiment, they have demonstrated the formation of novel complexes that cannot be achieved without the presence of the cage. Experiments using only the guest molecules did not result in a similar geometry as seen with the cages. The demonstration of variation is promising for the development of new “cube traps”, opening doors for us to explore otherwise impossible structures.
The type of chemistry used in this study draws on pre-existing concepts of steric hindrance, electronic compatibility and metal ligand interactions. The discovery of novel compounds is difficult to achieve without external assistance, as seen with the cube trap. This new approach, combined with the tunability of the cages, allows for better precision when searching for materials that can meet specific needs for various applications. This is an exciting step forward in inorganic chemistry and with worth additional study and exploration.

