Featured image and figures reproduced or adapted with permission. Further permissions related to this material should be directed to ACS.
Title: Spatially Resolved Production of Platinum Nanoparticles in Metallosupramolecular Polymers
Authors: Luis M. Olaechea, Lucas Montero de Espinosa, Emad Oveisi, Sandor Balog, Preston Sutton, Stephen Schrettl, and Christoph Weder
Journal: Journal of the American Chemical Society
Year: 2020
What if you could combine the advantageous properties of two materials just by mixing them? Trail mix combines the benefits of cashews and M&M’s, right? That is the idea behind the concept of nanocomposites, and specifically the incorporation of nanoparticles into polymers, two materials that are superstars of the chemistry world. Of course, even though combining two materials to make a “supermaterial” is a simple idea, in actuality its implementation requires careful design to address issues of compatibility between the two components and to be able to achieve the desired properties in the resulting material.
So why would you want to combine polymers and nanoparticles? Examples of the most basic uses of polymers can be seen in all the plastics surrounding the reader right now. A polymer can be tuned to modify its stiffness, flexibility, and strength to form materials as different as a plastic chair or Saran wrap. Nanoparticles, which are materials that are 1-100 nm in size, are also infamous for their tunability. In this case, simply altering their size can change their color and hence their optical properties due to the quantum confinement effect. They have a similarly wide range of uses including catalysis, where they can be used to facilitate the synthesis of useful molecules, and renewable energy, where they can be used to convert sunlight into electrical energy.
The ability to embed nanoparticles in a polymer broadens the scope of the applications that nanoparticles can be used for and allows them to be more widely incorporated into devices. For this reason, nanocomposites are important to the field of soft electronics, the technology that may give us incredible advances in personal electronics similar to the infamous flexible phone or even a phone screen printed directly on the body. The same technology has more practical uses in medical devices in the form of wearable diagnostic devices attached to the body like bandaids.

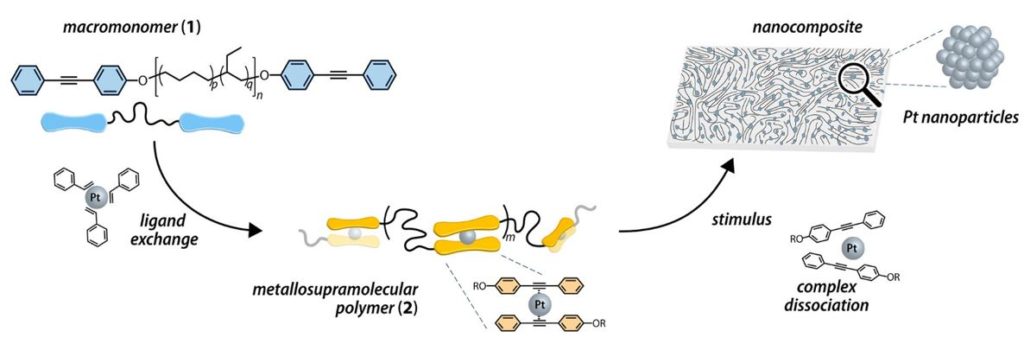
Researchers have recently developed a new method to produce a nanocomposite embedded with platinum nanoparticles. In short, the material can be made as an array of individually trapped platinum atoms in the polymer and upon the right stimulus such as light or heat, the platinum atoms coalesce to form nanoparticles. The same stimulus that forms the nanoparticles can be used to crosslink the polymer to make it stiffer. The resulting material, shown in Figure 1, is now a robust, free-standing sheet of dispersed nanoparticles ready for use in some device.
The monomer unit (compound 1 in Figure 2) that makes up this polymer is composed of long strands of poly(ethylene-co-butylene) (PEB) with diphenylacetylene end caps capable of binding platinum. In Figure 2, these end caps are shown as blue paddles connected on a string. When a solution of the monomer is mixed with platinum-bound styrene, an exchange reaction occurs between the styrene molecules and the monomer units because the platinum prefers to bind the monomer end caps (compound 2 in Figure 2) over the styrene. In the resulting nanocomposite precursor, each atom of platinum is held sandwiched between two alkyne groups of the diphenylacetylene end caps in a weak bond that can be broken by heat or light.
Researchers first demonstrated that platinum nanoparticles can be formed from this nanocomposite precursor by heating the material. Heating for 1 hour at 120 ℃ results in the formation of platinum clusters roughly 1 nm in diameter, as can be seen by the bright spots in the scanning transmission electron microscopy (STEM) images in Figure 3a. STEM can be thought of as an analog to conventional light microscopy except instead of focusing light on a sample, a beam of electrons is scanned across it to form an image with resolution down to the atomic level. Upon heating for a few days, a more ordered pattern can be seen in the STEM images, indicating that the clusters become more ordered themselves and are in fact crystalline (Figure 3b).

Finally, the most important implication of the unique synthesis of this nanocomposite is that its formation can be controlled by light. Shining light on a material in a controlled way is the go-to technique used by scientists and manufacturers to create increasingly small patterns, down to the nanoscale. This is because light can be focused down to an incredibly small spot the size roughly the wavelength of light, which for visible light is a few hundred nanometers. Using light to etch a pattern is called photolithography, which is the technique responsible for the production of the tiny computer chips present in our shrinking electronic devices.
Shining a UV light on the precursor, instead of heating the entire material, can selectively convert different areas of the precursor into the finished nanocomposite. This can be uniquely visualized using the catalytic properties of platinum nanoparticles. Light is applied to a single point on a sheet of the nanocomposite precursor, and when it is placed in a bath of hydrogen peroxide, bubbles of oxygen gas only form around the single pinpoint where the nanocomposite was converted (Figure 4). This is a simple test of spatial control, but it paves the way for customizable nanocomposites printed into intricate patterns.

