Title: Synchrotron-Based High Angle Resolution and High Lateral Resolution X-ray Diffraction: Revealing Lead White Pigment Qualities in Old Masters Paintings
Authors: V. Gonzalez, G. Wallez, T. Calligaro, M. Cotte, W. De Nolf, M. Eveno, E. Ravaud, M. Menu
Year: 2017
Journal: Analytical Chemistry
http://pubs.acs.org/doi/abs/10.1021/acs.analchem.7b02949
Art museums are great places to spend an afternoon admiring the magnificent works of past and current artists, but few people realize the extent to which scientists can incorporate high resolution analytical instrumentation to understand what exactly went into the creation of these masterpieces. That’s what a group of chemists from Paris, France set out to do when studying 14th-19th century Old European Masters paintings.
Paintings from this time period commonly employ a lead white pigment, which is composed of two distinct structural phases: cerussite PbCO3 (referred to as C) and hydrocerussite Pb3(CO3)2(OH)2 (referred to as HC). The lead white pigment was formed by suspending lead plaques above vinegar and exposing them to CO2 and heat (known as the “stack process”). C and HC then form in different ratios depending on the length of the exposure and the acidity of the synthesis environment. In order to determine what pigment qualities were used in the construction of 14th-19th century Old European Masters paintings and how they were related to production recipes, scientists from the Louvre and Sorbonne University used synchrotron radiation (SR)-based X-ray diffraction (XRD) to analyze microfragments from each painting.
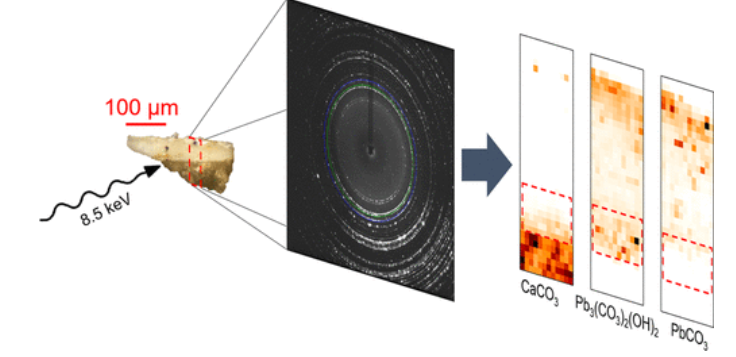
XRD, in general, uses high energy X-rays to determine the crystal structures and chemical compositions of thin films of material. Scientists can measure the scattered intensity of an X-ray hitting a sample, and different physical and chemical properties of the sample can then be determined depending on the incident and scattered angle, polarization, and energy of said X-ray. The scientists in this study took microfragment painting samples (samples less than 100 µm thick) to the European Synchrotron Radiation Facility in Grenoble, France to incorporate two distinct forms of SR-XRD. SR-high angular resolution (SR-HXRD) was used to determine the pigment composition, such as how much of the sample consisted of HC versus C, and to relate the crystal structure and size of the lead white pigment to its chemical properties. SR-high lateral resolution XRD (SR-µXD) achieved micrometric scale resolution in order to discriminate paint layers in a sample that was only several microns thick. This technique enabled the reconstruction of distinct structural phases for the entire sample in order to determine how each layer of lead white pigment varied in its distribution of HC and C.
The SR-HXRD measurements revealed that most of the lead white pigments obtained from the samples had a HC:C weight ratio of 60:40 or 80:20 (Figure 1). As described earlier, the synthesis of lead white pigment involves the exposure of lead to CO2. HC is the initial structural phase formed, but prolonged exposures to CO2 favor the conversion of HC to C. This transformation is extremely slow, therefore the scientists hypothesized that after HC was formed in sufficient quantities, the synthesis was stopped and the HC-heavy lead white pigment was collected. It is also historically known that there was a high demand for lead white paint during these time periods, so production rates may have been increased and synthesis corrosion times curbed to maximize the presence of HC. Some C rich samples may have been exposed to a post synthesis acid treatment, as C is favored to form over HC in acidic environments.
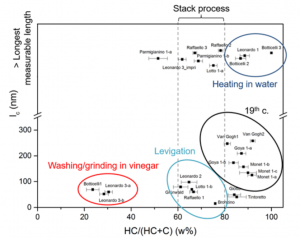
Figure 1: Structural size distributions and weight percentages of lead white pigment samples obtained from 14th-19th century Old European Masters paintings.
The scientists also successfully correlated the weight percents with the microstructures and lengths of the crystals present in the lead white pigment samples (Figure 1). The pigments that contained a low percentage of HC (and therefore were rich in C) consisted of smaller sized particles, supporting the hypothesis that a post synthesis acid treatment in vinegar could contribute to the recrystallization of small C microstructures. Levigation could have decreased the particle size of the lead white pigment without changing the 60:40 HC:C ratio, while heating the pigment in water could account for a larger particle distribution in the 80:20 and 90:10 H:C ratio regime.
Finally, the researchers chose two samples (Beaumetz and Bellechose) to create SR-µXD crystalline maps for to determine the structural differences between distinct microlayers of lead white pigments. Figure 2 illustrates the phase maps of each sample and reveals their stratification of calcite, C, and HC. The Beaumetz sample consisted of a preparatory layer of calcite (1A in Figure 2), followed by a priming layer of HC (2A) and a pictorial layer mixing HC:C in a 75:25 ratio (3A). The Bellechose sample had more complex HC:C ratios in the lead white layers, ranging from 80:20 to 90:10 depending on the layer. The SR-µXD information supports the conclusion that a reduced price blanc de puille white pigment was the primary color used in these paintings.

Figure 2: SR-µXD mapping of calcite, C, and HC for samples Beaumetz (A) and Bellechose (B).
This study revealed the often unknown interplay between high resolution analytical instrumentation and art history. By using SR-based XRD analytical configurations, these researchers identified chemical composition ratios, pigment crystallite sizes, and stratification of paint layers on the microscale from samples obtained from 14th-19th century Old European Masters paintings. The chemistry revealed in these studies can be connected to the grades of lead white pigments used by painters, as well as to the different synthesis conditions or post synthesis treatments employed by paint manufacturers and artists of the past. Next time you are wandering around an art museum, make sure to stop to appreciate the vast level of analytical detail available about a painting if you use the proper technique.