Featured Image Source: https://calmatters.org/wp-content/uploads/sites/2/2020/02/Coronavirus_illustration.jpg
Primary Source Info:
Title: The Antiviral Compound Remdesivir Potently Inhibits RNA-Dependent RNA Polymerase from Middle East Respiratory Syndrome Coronavirus
Authors: Calvin J. Gordon, Egor P. Tchesnokov, Joy Y. Feng, Danielle P. Porter, Matthias Götte
Year: 2020
Journal: Journal of Biological Chemistry
DOI: http://dx.doi.org/10.1074/jbc.AC120.013056
The 2019 novel coronavirus SARS-CoV-2, the virus that causes the disease COVID-19, is a recently emerged pathogen that has rapidly spread across the world and now represents the single most significant public health challenge globally. SARS-CoV-2 originated in bats and is believed to have been hosted in pangolins before transmitting to humans1. Like all coronaviruses, SARS-CoV-2 most frequently causes respiratory disease, although in severe cases damage to other organs including the heart and kidneys is also observed. As there are no drug treatments currently available for COVID-19, academic labs and the biopharmaceutical industry have been rapidly searching through known bioactive compounds to identify any that can inhibit the progress of the virus or otherwise reduce the severity of the disease.
The viral cycle of SARS-CoV-2 begins with binding of the viral spike (S) protein, which binds to a human receptor protein on the surface of some cells called ACE2. The ability to effectively interact with human ACE2 is believed to be the major alteration that allowed the virus to “jump” from previous animal hosts to humans. After binding ACE2, the virus fuses with the membrane of the human cell, releasing its internal contents into the cytosol (the main interior compartment of the cell). This payload includes the viral genome as well as several other proteins necessary for the virus to replicate. One of these proteins, an enzyme called RNA-dependent RNA polymerase (RdRp), is responsible for making copies of the viral genome (Fig 1).
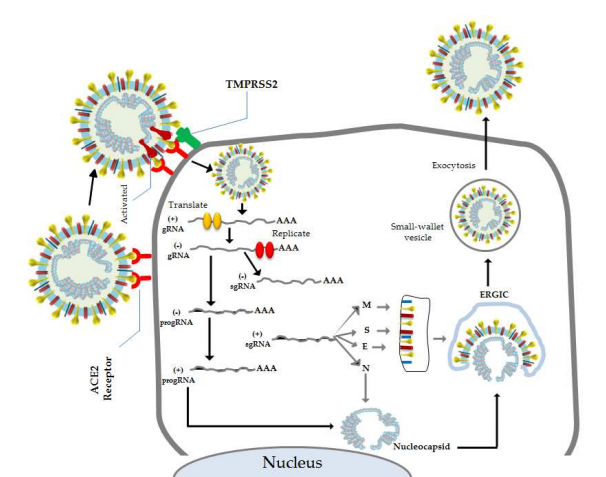
Figure 1: Coronaviruses first bind to the cell surface (left) where they inject their genetic material to be replicated and then translated into viral proteins (bottom). These proteins then envelope a newly synthesized viral genome and bud off to infect other cells (right). Image Source: Astuti and Ysrafil, 2020.
Viruses can carry their genetic information on either DNA or RNA, and either of these can be single-stranded or double-stranded. Any double-stranded nucleic acid has one positive-sense strand, which is read by the cell’s protein synthesis machinery, and one negative-sense strand, which is complementary to the positive-sense strand. Single-stranded viruses may carry either the positive- or negative-sense strand as their genetic material, since either of these contains sufficient information to replicate its complement. Coronaviruses fall into a category of viruses known as single-strand positive-sense RNA viruses. This means that they carry their genetic information on single-stranded RNA (as opposed to DNAor double-stranded RNA, for example), and that this RNA directly encodes the viral proteins (positive-sense). The RdRp that gets injected to the cell along with the coronavirus positive-sense genome is first replicated by RdRp to create a complementary negative-sense template. Then, this template is used to create many copies of the positive-sense strand, which hijack the cell’s protein synthesis machinery to produce new viral proteins. These proteins self-assemble into viral particles, which bud out from the cell and go on to restart the infectivity cycle2.
One class of drugs that has been successful in fighting off other viral infections, like HIV, has been nucleotide analogues. These drugs are chemically similar to the nucleotides that cell’s use as the building blocks of nucleic acids, DNA and RNA, which make up genetic material. Because these analogues are similar to nucleotides, they can block the activity of enzymes that synthesize nucleic acids. As a result of their past successes, it is not especially surprising that one of the first drug candidates to be approved for use in COVID-19 is a nucleotide analogue: remdesivir (Fig. 2). Remdesivir was developed by the biopharma company Gilead as a treatment for Ebola virus, but it was found to have antiviral activity against many different viruses. While it was safety-tested in humans, it was not taken through all of its trials because more effective treatments for Ebola had already been developed. Nonetheless, promising early reports prompted the investigation of remdesivir in COVID-19 therapy.
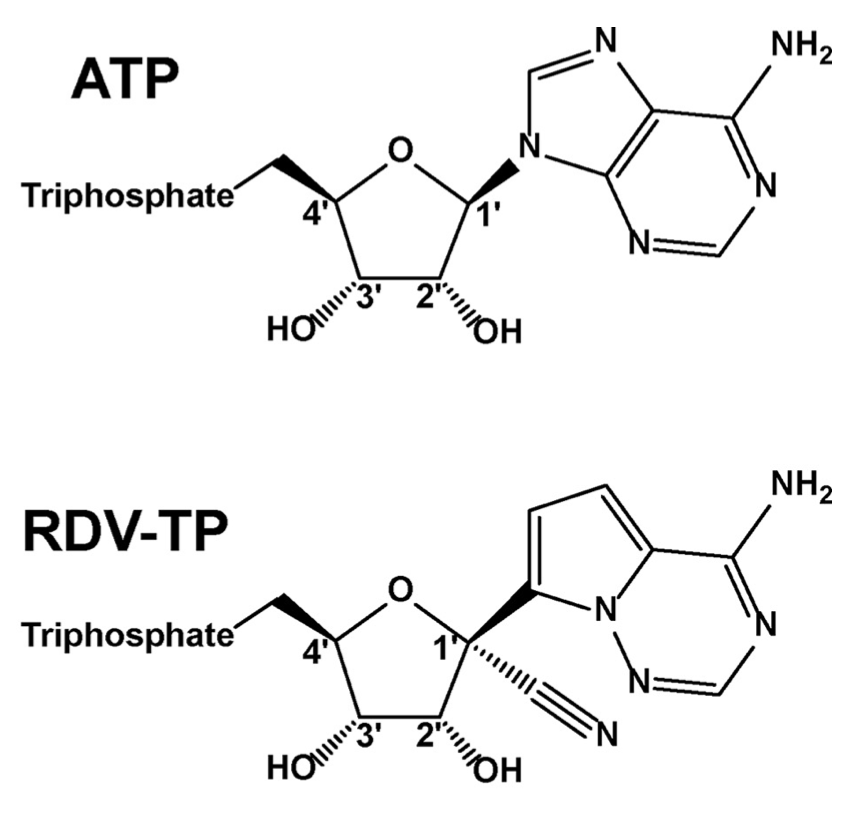
Figure 2: Chemical structures of ATP (top) and the ATP analogue remdesivir (bottom).
Previous research on the effect of remdesivir on RdRps of other viruses focused on Ebola and other single-strand negative-sense RNA viruses – these viruses carry the opposite strand as coronaviruses, and must replicate the complement of their genome for use in protein synthesis, making their life cycle slightly different. To explore remdesivir’s activity against coronaviruses, Calvin Gordon at the University of Alberta partnered with scientists at Gilead to confirm the ability of remdesivir to inhibit RNA polymerization by a coronavirus RdRp. Alongside confirming the efficacy of remdesivir against the RdRp in SARS-CoV-2, these experiments answer an important question in why remdesivir, specifically, can successfully inhibit coronavirus RdRps while many other nucleotide analogues cannot. One interesting feature of the coronavirus RdRp is its proofreading 3’ exonuclease. Most viruses do not bother to proofread their genome when they replicate, which accounts for the rapid evolutionary change observed in viruses like the flu and HIV. While this means that coronaviruses mutate less rapidly than other viruses, it also poses a challenge for the development of nucleotide analogues: if the enzyme detects these analogues as a “mismatch” once they are incorporated, the proofreading exonuclease on RdRp will chop it off and incorporate the regular nucleotide in its place, thus rendering the drug ineffective. Understanding the role of the proofreading exonuclease in remdesivir inhibition is therefore crucial to the success of remdesivir as a COVID-19 therapy.
To answer these questions, the authors performed biochemical experiments to measure RNA replication by RdRp in the presence or absence of remdesivir. They used RdRp from the MERS coronavirus, which is very similar to SARS-CoV-2, and gave it a template from which to synthesize a new RNA strand (Fig. 3). They tracked the synthesis of a new RNA polymer by incorporating a radioactive nucleotide and performing gel electrophoresis. Gel electrophoresis is a common molecular biology technique where charged molecules, such as RNA, are loaded into a gel and an electrical current is applied. This causes the RNA to move through the gel at a rate inversely proportional to its mass. In this way, RNAs of different sizes can be separated, while the radioactive label enables the RNAs to be visualized.
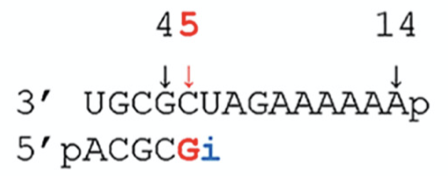
Figure 3: The authors used a 14 nucleotide template (top) to track new RNA (bottom) synthesis by RdRp. The radioactive nucleotide is incorporated at position five (red). The authors can be certain that remdesivir will incorporate at position 6 (blue i) because remdesivir is an A analogue, and A pairs with U in RNA.
The authors observed that remdesivir effectively stalls RNA synthesis by RdRp at therapeutically attainable concentrations (Fig. 4). They also quantified the enzyme’s preference for remdesivir versus the regular nucleotide it mimics, ATP. Interestingly, while most nucleotide analogues are incorporated less efficiently than their normal counterparts, remdesivir actually outcompetes ATP for incorporation, making it a highly effective inhibitor. Another unique feature of remdesivir was where the chain was terminated – if remdesivir is incorporated by RdRp at position i, the resultant chain had a length of i + 3, meaning the enzyme went on to incorporate three more nucleotides before stalling. This means that remdesivir is no longer available to be excised by the proofreading exonuclease, and it explains why remdesivir seems to be effective against coronaviruses when other nucleotide analogues might not be.

Figure 4: The authors gel electrophoresis data demonstrating remdesivir inhibition. The farther a band runs down the gel, the longer the RNA strand is. From left to right, the authors incorporate increasing concentrations of remdesivir (RDV-TP). As the concentration of remdesivir increases, the band corresponding to the completed strand (red box) disappears, and a smaller band (blue box) takes its place, corresponding to an RNA of length i+3.
Remdesivir is currently in a number of Phase III clinical trials (the final phase before approval) for the treatment of COVID-19. The first US trial released its results earlier this week, and the FDA is currently in the process of granting its approval. While these signs are encouraging, significant challenges with remdesivir remain. For one, many antiviral drugs, including remdesivir, must be given early in the course of disease to be effective, before the virus has the opportunity to “take-hold”. This is also true of the anti-flu drug Tamiflu, for example. Additionally, remdesivir is challenging to synthesize: even under the best of circumstances, there will probably be fewer than two million courses available globally before 2021. As a result, it will be important to ration its use for patients at the highest risk of developing severe disease. Nonetheless, for a disease without any available drugs yet, approval of remdesivir for the treatment of COVID-19 has real potential to reduce the burden on our healthcare system and the fatality rate of the disease.
Other References:
- Boni, M. F., Lemey, P., Jiang, X., Lam, T. T.-Y., Perry, B., Castoe, T., Rambaut, A., & Robertson, D. L. (2020). Evolutionary origins of the SARS-CoV-2 sarbecovirus lineage responsible for the COVID-19 pandemic. BioRxiv, 2020.03.30.015008. https://doi.org/10.1101/2020.03.30.015008
- Astuti, I., & Ysrafil. (2020). Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. https://doi.org/10.1016/j.dsx.2020.04.020

