| Title: | Tuning Single Quantum Dot Emission with a Micromirror |
| Authors: | Gangcheng Yuan, Daniel Gómez, Nicholas Kirkwood, and Paul Mulvaney |
| Publication Info: | Nano Letters, 2018, Article ASAP DOI: 10.1021/acs.nanolett.7b04482 |
Everyone’s familiar with mirrors. You probably look in one every morning: check out how great your hair looks today, maybe use it to take a rad selfie. But the idea of using mirrors to look at molecules – that just sounds crazy, right? In fact, a groundbreaking work in the 1940s was the first to use a tiny mirror to look at the fluorescence emission of a single molecule. Scientists today continue to build on this work. In this Chembite, we’ll talk about how some researchers used this tiny-mirror method to look at one of today’s most exciting new materials: quantum dots.
If the name “quantum dot” is bringing back memories of physical chemistry class, you’re on the right track. Quantum dots are tiny, roughly spherical particles, often of an inorganic crystal like cadmium selenide (CdSe). Because they’re so small, they have some pretty interesting properties. One of these is that small differences in a quantum dot’s size can directly affect the wavelength of light that it absorbs and emits. This makes them ideal for applications like light emitting diodes (LEDs). You may even have seen quantum dot TVs advertised – but they have many applications outside your living room as well.
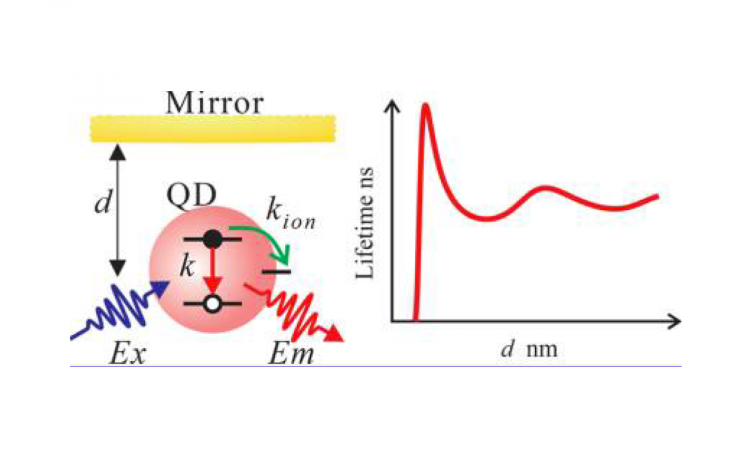
One major obstacle facing quantum dot technology is a phenomenon known as “blinking.” After a quantum dot absorbs light, it must get rid of that energy somehow. One way is to re-emit this energy as light. The process by which the quantum dot re-emits this light enables technologies like LEDs and is called fluorescence. But in “blinking” quantum dots, sometimes this emission of light won’t occur because the energy has gone into a competing process in the quantum dot. Understanding why blinking occurs, as well as how to measure the amount of light a quantum dot is emitting despite this blinking, brings us to the micromirror employed by researchers from Paul Mulvaney’s group at the University of Melbourne in Australia.
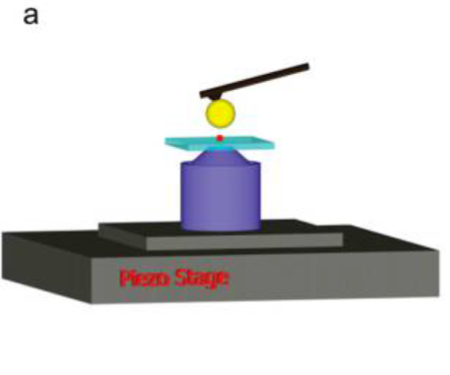
The gold micromirror employed by the researchers is really a sphere, but since it is so much bigger than a single quantum dot, it can be approximated as flat. The researchers glued it onto the tip of an atomic force microscope (AFM), which allows them to accurately bring the mirror within nanometer distances of their target. The quantum dots themselves are embedded in a polymer matrix on glass so that a laser can be directed onto the quantum dots from the other side. The laser “excites” the quantum dots to a higher energy; this energy can then be emitted by the quantum dots as fluorescence. The mirror can move back and forth across the quantum dot, reflecting the intensity of its fluorescence back to a detector. As the quantum dot “blinks,” the intensity of the fluorescence fluctuates between “on” and “off” states over time. That’s pretty amazing given that the quantum dot is only a few nanometers across, and the mirror is within 1000 nm of the quantum dot’s surface throughout the measurement! (To give you an idea of just how small a distance that is – a human hair is 75 times that distance in diameter.)
The researchers here were specifically interested in seeing if the mirror itself affects the emission of the quantum dots. In order to look at this, they brought the mirror too close, so that it actually contacted the film (within 20 nm of the center of the quantum dot). They also fully retracted the mirror, so that it was far from the surface of the quantum dot. They looked at the amount of light emitted by the quantum dot in each scenario in order to measure the mirror’s effect and found that although the mirror does not change the “off” periods of the quantum dot blinking, it does lengthen the period of the “on” times. They attribute this lengthening to the mirror enhancing the rate of fluorescence of the quantum dot, which means that the quantum dot becomes more likely to emit light than to undergo the competing processes leading to blinking. However, because the mirror only lengthens the amount of time spent in the “on” state, and doesn’t change any of the other measurements, it’s still a great way to accurately measure the amount of light emitted by these quantum dots. That makes it a good technique for any researchers trying to optimize LED technology.
Looking at individual molecules – or quantum dots – is a pretty tricky process. Just imagine trying to isolate something much, much smaller than even a grain of sand – let alone measure any of its properties! So make no mistake, this is no ordinary mirror. Instead of helping you out with your selfie, it’s helping researchers paint a picture of chemistry on a nanometer scale.