Authors: Kai Zhang and Yuegang Zuo
Journal: Journal of Agricultural and Food Chemistry
Affiliation: Department of Chemistry and Biochemistry, University of Massachusetts Dartmouth
| Figure portion reprinted with permission from J. Agric. Food Chem., 2004, 52 (2), pp 222–227. Copyright 2004 American Chemical Society. |
Does your mental picture of Thanksgiving include phenolic acids? Perhaps it should!
We rarely think about chemistry in relation to food, especially given the stigma of “chemicals” in food, but chemicals constitute the nutritional value, the delicious (or not-so-delicious) flavors, and arguably everything else about what we eat. Chemistry can therefore play a valuable role in understanding the way we interact with food, both in terms of tastes and textures and in terms of biology.
To do the biological study of how particular (good) chemicals like antioxidants affect people, there needs to be a quick, reliable, and consistent way to measure those chemicals in humans. Furthermore, a good method would test for multiple chemicals, because the health benefits from some chemicals only exist in the presence of others (phenolic acids and flavonoids in this case). The authors of this paper demonstrate a gas-chromatography mass-spectrometry (GC-MS) method to investigate how some of the chemicals present in cranberries (from cranberry juice) go into your bloodstream after you ingest them.
Zhang et al. performed an ethyl acetate extraction on human blood plasma samples taken 45 minutes and 270 minutes after the subjects consumed cranberry juice. The phenolic and benzoic acid compounds in the ethyl acetate phase are allowed to react with a trimethylsilyl-ating reagent (BSTFA (N,O-bis(trimethylsilyl) trifluoroacetamide) + TMCS (trimethylchlorosilane)) so that the derivatives can be easily identified as peaks in the GC-MS spectrum.
The ever-important control samples to qualify this technique as legitimate were samples of the subjects’ blood plasma before the consumption of the cranberry juice, and also samples of the pre-cranberry blood with the intentional addition of a known amount of 20 standard phenolic compounds. The latter samples allow the experimenters to determine to what extent their method is able to quantitatively determine levels of these various compounds in the same environment as the real post-cranberry samples. They found that the ethyl acetate extraction was able to achieve at least 63% recovery for the standard phenolic compounds, with the exception of o-phthalic acid, which had a recovery of 30% that is presumably due to its low solubility in ethyl acetate or a strong binding affinity toward components of the plasma.
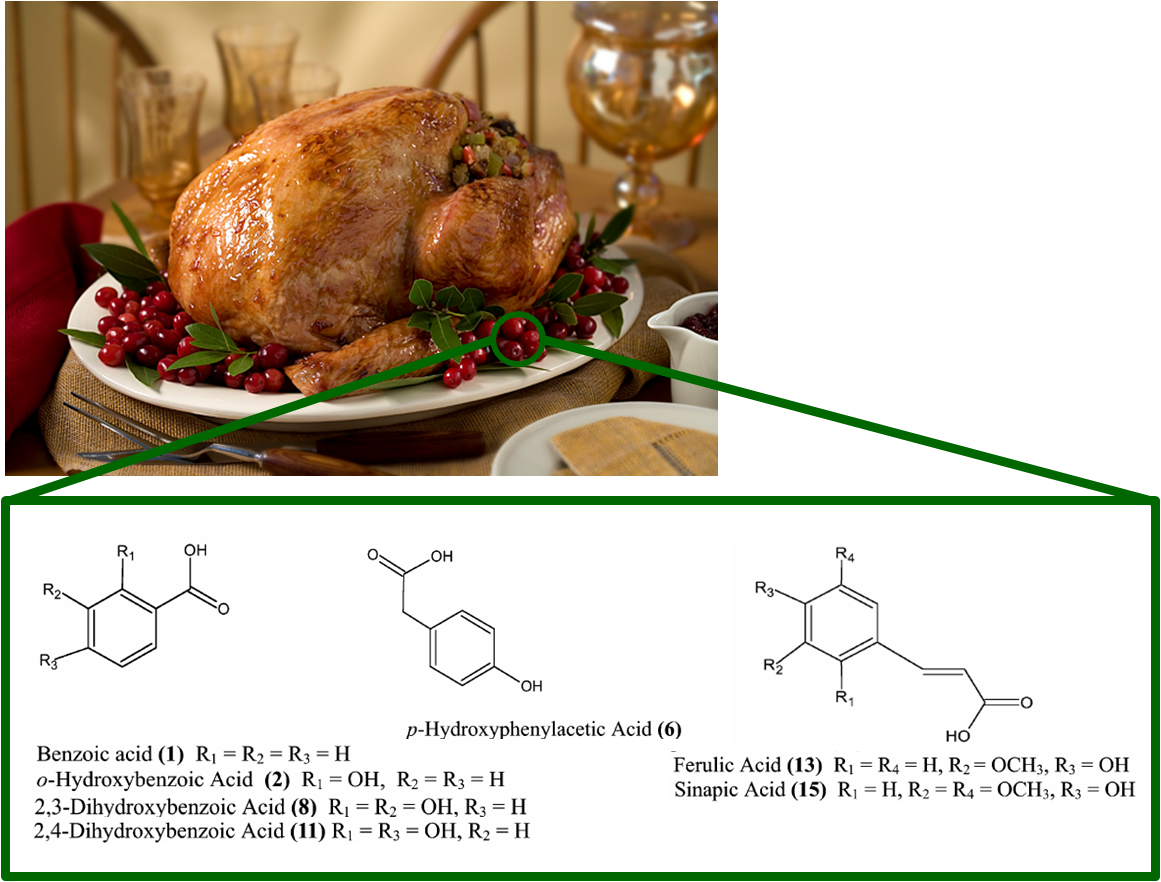
The compounds found in the 45-minute samples were benzoic acid (1), o-hydroxybenzoic acid (2), p-hydroxyphenylacetic acid (6), 2,3-dihydroxybenzoic acid (8), and 2,4-dihydroxybenzoic acid (11) (numbers indicate the chemical structures from the figure). The 270-minute samples contained these compounds as well as ferulic acid (13) and sinapic acid (15). One interesting point about these compounds is that two of them (p-hydroxyphenylacetic and 2,4-dihydroxybenzoic acid) are not found in the cranberry juice, but they are found in the human blood plasma after consuming cranberry juice (and not before). This result suggests that these two compounds are the result of chemistry occurring in the human body that uses some of the original phenolic compounds in the cranberry juice as reactants!
This simple extraction and GC-MS method can be used for a variety of compounds-not just those in cranberries. The authors hope this method will be useful in measuring levels of antioxidants in biological studies to determine their bioavailability and pharmacokinetics (how the body processes the chemicals it ingests).
Happy Thanksgiving from chembites!