Authors: Nikita Hanikel, Mathieu S. Prevot, Frahad Fathieh, Eugene A. Kapustin, Hao Lyu, Haoze Wang, Nicolas J. Diercks, T. Grant Glover, and Omar M. Yaghi
Journal: ACS Central Science
Year: 2019
Article: https://pubs.acs.org/doi/pdf/10.1021/acscentsci.9b00745
Water scarcity plagues many regions of our planet, especially in arid environments. This issue is likely to get even worse as the climate changes and extreme heat days occur more frequently in many places. Additionally, a growing population requires even more water to grow enough food for everyone. However, these researchers have invented a new way to capture water from air using chemistry!
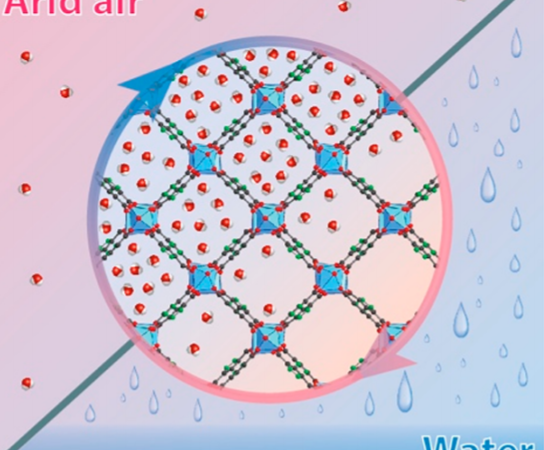
Previous challenges in this field have come from the fact that adsorption of the water usually happens at night and then the water is released from the membrane during the day (Figure 1). The membrane is made of a metal organic framework (MOF) and is very porous so has the ability to absorb gas molecules inside the structure of the atoms. An image of this structure can be seen in Figure 2. This process doesn’t produce enough water to be useful, so these researchers here have developed a way to generate multiple cycles of water harvesting per day.
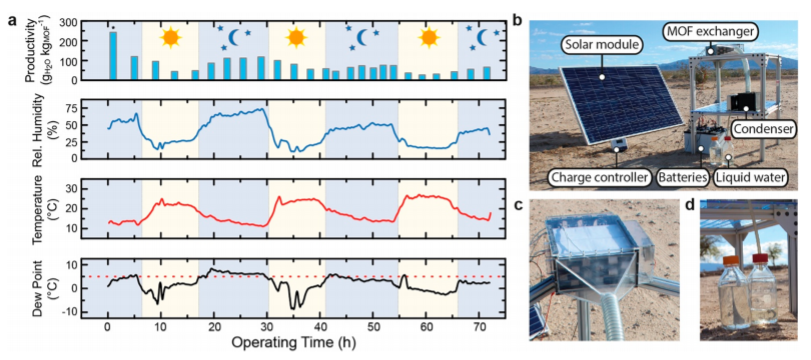
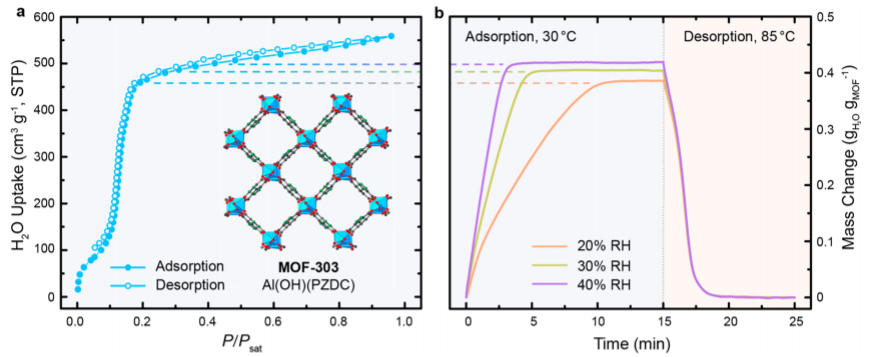
They have developed a new MOF containing aluminum that outperforms other materials for water uptake. They measure the water uptake over time at different relative humidity values (Figure 2). Nine water harvesting cycles are able to be completed in each day and there is no decrease in water absorption after 150 cycles. They find that in an indoor arid environment at 32% relative humidity, the system can absorb 1.3 L of water per kg of MOF per day. This technology can even operate in the Mojave desert and still collect 0.7 L of water per day. This is particularly exciting because the dry regions are most likely to suffer from lack of water. Moreover, this can be implemented in remote locations without electricity.
To demonstrate the viability of this method in a dry setting, researchers took the instrument into the field to collect samples for three days (Figure 3). They were able to run the instrument in an extremely dry climate and collect a significant amount of water running only on solar power. This technology will revolutionize the way people around the world cope with water stress and will help us adapt to a growing population and a warming planet.