Title: Renewable carbon feedstock for polymers: environmental benefits from synergistic use of biomass and CO2
Authors: Marvin Bachmann, Arne Kätelhön, Benedikt Winter, Raoul Meys, Leonard Jan Müller, and André Bardow
Year: 2020
DOI: 10.1039/d0fd00134a
Polyurethane is a polymer that’s commonly used to produce foams and hard plastics. Polyurethane, like many plastics, is made from side products of fossil fuels including oil and coal. As the world moves towards more renewable energy sources, alternative sources need to be found to produce polyurethane and other polymers.
Recently, researchers from Germany and Switzerland studied the possibility of replacing fossil fuel feedstocks with a mix of biomass and carbon dioxide. Biomass is carbon-based building block chemicals obtained from crops (in this paper the researchers studied a type of grass called miscanthus). Carbon dioxide, alternatively, can be turned into building block chemicals through a variety of different chemical processes. Both these methods limit carbon dioxide emissions that contribute to global warming because plants draw carbon dioxide out of the atmosphere during their growth and the carbon dioxide itself is removed from the atmosphere to make the building-block chemicals.
The researchers identified that while using carbon dioxide or biomass avoids the carbon emissions associated with using fossil fuel feedstocks, there are also drawbacks to consider. Capturing carbon dioxide requires energy. If this energy is supplied in the form of fossil fuels, it causes carbon emissions itself and if renewable energy is used, then that electricity can’t be used elsewhere. Biomass needs to be grown on farms and there are potential carbon emissions from the farming process or from changing forest or grassland to cropland. The researchers reasoned that minimal carbon dioxide emissions and environmental effects may be achieved by using a mixture of biomass and carbon dioxide starting materials.
To analyze the impact of polyurethane synthesis, the researchers performed a life cycle analysis. In a life-cycle analysis, the impact of a product at each stage of its life, from manufacturing through use and eventual disposal, is calculated. They found that using carbon dioxide captured from the atmosphere in the production of polyurethane could decrease the carbon emissions from 7 kg CO2 for 1 kg of polyurethane to between four and 4.5 CO2/kg polyurethane if the electricity used in the process came fully from renewable sources. Using biomass could decrease emissions in polyurethane production from 7 to just over 4 kg CO2/kg polyurethane, although this would come at the cost of needing to change the land currently used for other purposes to farming biomass crops. Combing both biomass and carbon dioxide as building blocks could cut the emissions further to 3.1 kg CO2 eq/kg of polyurethane (Figure 1). This further decrease is because carbon dioxide is produced in the process of converting biomass to starting materials for polyurethane synthesis and that carbon dioxide can be conveniently used to make other starting materials with minimal extra emissions.
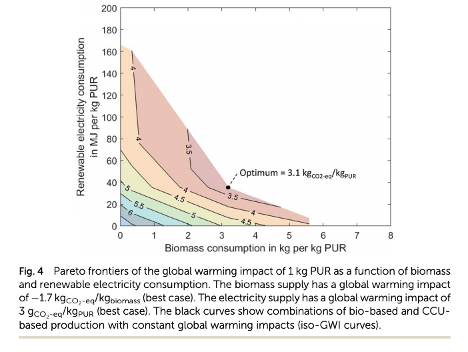
Figure 1: The black lines represent different carbon dioxide emissions for the synthesis for the synthesis of polyurethane (in grams of carbon dioxide/kilogram of polyurethane). The minimal carbon emissions are 3.1 kg of carbon dioxide per kg of polyurethane using a mix of biomass and carbon dioxide as starting materials (Image used under creative commons license).
The authors note that while combining carbon dioxide and biomass feedstocks can lower the emissions of making polyurethane, there are other environmental parameters that can be harmed by the combination. Both processes increase the use of metals and other minerals as well as the presence of air pollutants that can be damaging to human health (Figure 2). Additionally, both methods release more acid into local land and water than using fossil fuel starting materials do. The authors note that these parameters must be considered alongside the carbon-emissions reduction when evaluating the best production methods for polyurethane.
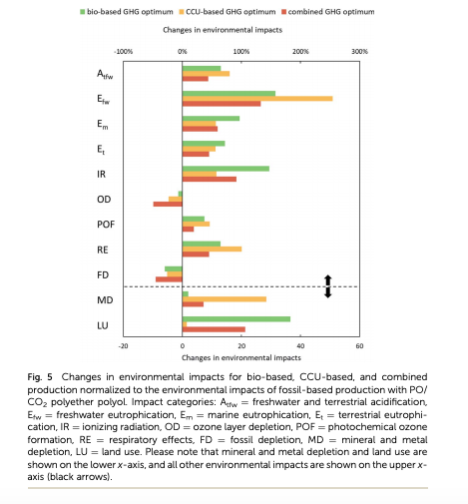
Figure 2: While the combined feedstock method decreased carbon emissions relative to using fossil fuels it increased 9/11 environmental parameters the researchers examined relative to using fossil fuel feedstocks (Used under creative commons license).
Finally, the authors note that the starting materials used for polyurethane synthesis are also common in the production of many other polymers and chemical products. Future work could focus on the emissions and environmental impact of performing these processes starting from biomass or carbon dioxide feedstocks as well as examining how multiple processes starting from those materials might affect the practical and environmental issues involved in polymer synthesis.
