Title: Simultaneous analysis of multiclass antibiotic residues in complex environmental matrices by liquid chromatography with tandem quadrupole mass spectrometry
Authors: Azhar Rashida, Sohaib H. Mazharc, Qiaoting Zenga, Claude Kikia, Chang-Ping Yua, Qian Sun
Journal: Journal of Chromatography B
Year: 2020
DOI: 10.1016/j.jchromb.2020.122103
Don’t panic! The paper won’t hurt you… Sometimes, we all look at a paper and feel too overwhelmed, not knowing where to start. Well, hopefully I can help with that. First, I’d like you to reread the title. “Simultaneous analysis of multiclass antibiotic residues in complex environmental matrices by liquid chromatography with tandem quadrupole mass spectrometry”. That does sound complicated… So, let’s break it down! What do we know? Well, “liquid chromatography with tandem quadrupole mass spectrometry” is an analytical technique, commonly used in environmental chemistry to detect and quantify a huge variety of compounds. It’s a favourite technique of researchers in this field as it has the ability to detect and quantify very low concentrations of compounds (known as the “limit of detection – LOD”, or “limit of quantification – LOQ”), which is especially important when considering the “complex environmental matrices” being studied! The Matrix is a trilogy starring Hugo Weaving, and is… Wait, what was I saying? Ah, that’s the wrong matrix! When a paper like this refers to “environmental matrices”, they mean the external surroundings in which a compound “lives”. This matrix normally affects how the compound behaves, for instance, does it get stuck in the soil or flow out in the water? In this case, the matrices are chicken manure, swine manure, poultry feed, sediment and soil samples. These matrices normally contain numerous compounds, and Researchers tend to only want to see one (or one group of) compound(s) at any one time, and these matrices normally contain numerous compounds, so they are considered “complex”. Therefore, being able to detect and quantify low concentrations amongst the rest of the random stuff in the matrix is vital. See the “Antibiotics in our food?” blog post (written by yours truly) for more information on how LC-MS works…
The title here also tells us what they are detecting – “multiclass antibiotic residues”. We all know what antibiotics are. The “multiclass” bit, however, is not so well-known. Antibiotics come in a variety of shapes and sizes. These different groups are called “classes”. If all antibiotics were the same, it would be very easy for bacteria to learn how to defend against them. Having this variety, therefore, means that if bacteria become resistant to one type of antibiotic, we can use a different antibiotic from a different class! Each class works in different ways, for example, oxytetracycline (in the tetracycline class of antibiotics) works by preventing the bacteria from producing essential proteins, while metronidazole (imidazole class) attacks and destroys the DNA. These different attacking mechanisms are known as the “mode of action”, and each different class has its own unique “attacking” style (think the different fighting styles of Gimli and Legolas!). Returning to the title, “simultaneous analysis of multiclass antibiotic residues”, we can understand that the researchers are analysing multiple antibiotic classes at the same time. Now we understand the title, let’s jump in!
The overall aim of this paper was to develop an appropriate method using LC-MS that could quantify the concentrations of 40 different antibiotics at the same time. This is no easy task! Due to the chemical and biological properties being so diverse across these 40 antibiotics, they each require different analytical conditions, and so finding one method that works for all is certainly a challenge.
Prior to analysing any real samples, the researchers tested out several different methods with spiked samples (i.e. blank samples spiked with known concentrations of the antibiotics). They obtained data from each method, and from this they could calculate percentage recoveries. It’s important to note that the percentage recovery can be greater than 100% – one potential cause for this is ion enhancement, i.e. something is making the ion peak larger in the mass spectrum than it would otherwise be. Calculating the percentage recoveries allows the researcher to identify the most appropriate method for each antibiotic, and thus choose the one that performs the best for the most.
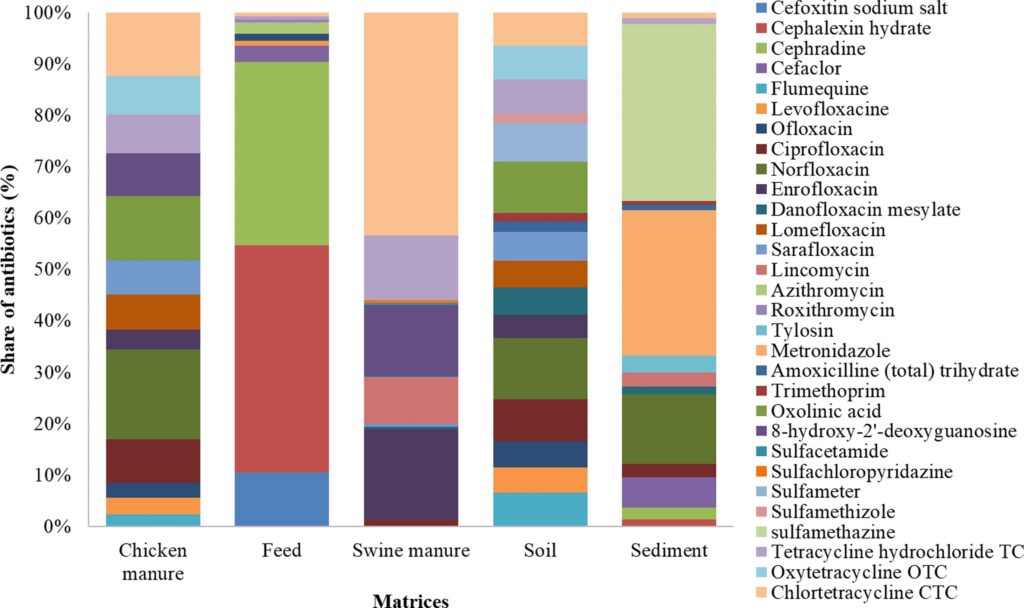
Once the method has been chosen, the samples are cleaned up to remove the dirt and the antibiotics are extracted. After extraction and a few more complicated technical calculations (e.g. the LOD and LOQ), it’s time for the analysis! Rashid et al found that all the matrices contained antibiotic residues. Chicken manure, swine manure and soil contained much higher total amounts of antibiotics than sediment and poultry feed. These were all detected with the residues of fluoroquinolone, macrolide, ulfonamide and tetracycline antibiotics.
Figure 1 shows the distribution of different antibiotics in the different matrices. This shows that the antibiotics were detected at different concentrations in the different matrices, for example, chlortetracycline was discovered at a concentration of 196 µg/kg in swine manure, compared to 88 µg/kg in poultry feed and between 44 and 46 µg/kg in soil. This suggests that antibiotics stick around in different matrices to different extents, and therefore that looking at only one matrix when analysing antibiotics in the environment is not enough.
Papers like this are vital to research life – they are commonly referenced and used to develop more methods. If somebody else was wanting to study one of the same antibiotics, instead of developing a brand-new method, they might want to read this paper to get an idea of limits of detections, what solvents would/wouldn’t be good, and whether they could improve on what is already published. Learning how to read such papers, and pick out the relevant information, is therefore incredibly important!
Disclaimer: Figures and articles are re-used purely for academic purposes with no intent of profit.

