Authors: Phillip Crews, Paul Dorenbach, Gabriella Amberchan, Ryan F. Keiffer, Itzel Lizama-Chamu, Travis C. Ruthenburg, Erin P. McCauley, and Glenn McGourty
Journal: Journal of Natural Products
Year: 2022
Featured image by Jim Fischer is licensed under CC BY 2.0 by Creative Commons.
With an increase in wildfire frequency the last few years, wine experts fear that higher smoke levels will ultimately lead to a decline in wine quality from grapes cultivated in these areas. While grapes and vines tend to survive smoke damage from distant wildfires, there has been little research on the potential effect on quality of grapes harvested after wildfire exposure in American vineyards. This study aimed to expand on research pioneered by the Australian Wine Research Institute, responsible for decades of work looking into the effect of smoked grapes on eventual wine flavor and quality.
Smoke-exposed grapes are damaged by the degradation of plant polyphenolic compounds. When the plants are exposed to high heat, polyphenolic compounds within the plant are broken down into volatile phenols. While odorless, these volatile phenols have been previously shown to have a negative correlation with wine quality; the more volatile phenols are present, the lower the quality of wine. Wood-derived smoke also includes over 500 additional volatile phenols, resulting in even more phenol contamination. Previous efforts to quantify volatile phenols in wine samples include the direct quantification of free volatile phenols via GC-MS1 as well as detection of the total volatile phenols in a sample via hydrolysis followed by detection.2,3 However, these methods are not the most efficient markers of quality as levels of unbound volatiles tend to be very low and not representative of the sample and detection of total volatiles requires indirect determinations after the volatile phenols have undergone a chemical transformation.
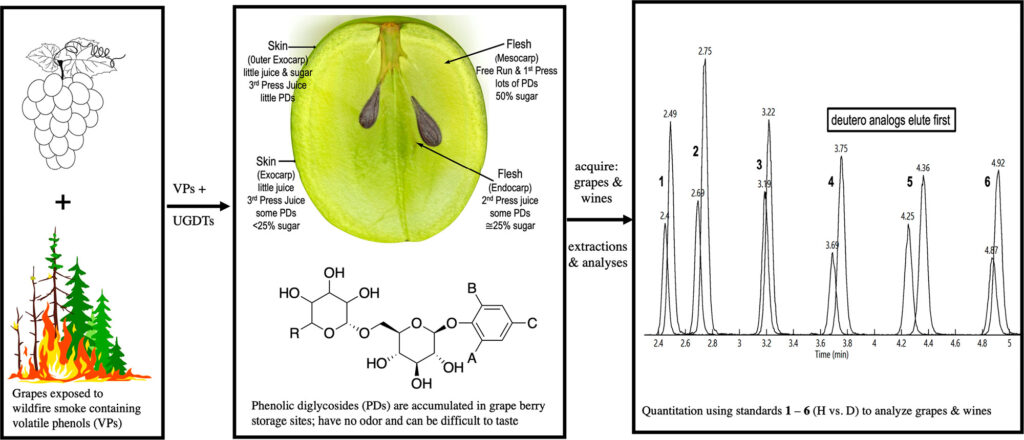
To address these detection limitations, Crews et al. developed a method to directly detect bound, glycosylated volatile phenols (PDs). Volatile phenols diffuse through grapes, spreading to various storage sites in the berry where they are transformed into bound PDs by various enzymes naturally occurring in the fruit. Conveniently, PDs such as these are available as commercial standards, making it easy to both identify and quantify bound PDs in various samples via mass spectrometry (Figure 1).4
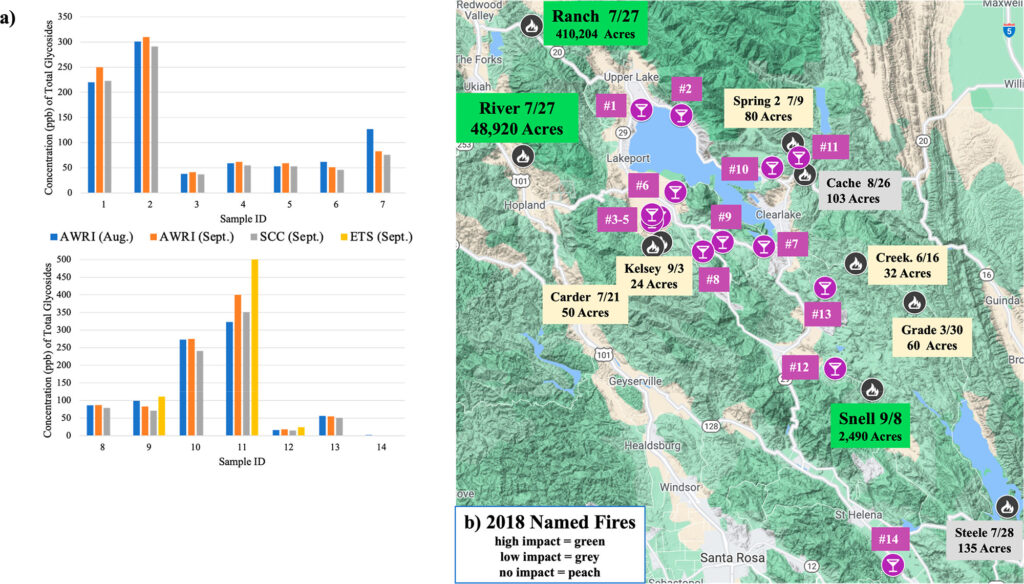
A 2018 harvest of Cabernet Sauvignon was one of the main focuses of this study. Out of fourteen vineyards with varied levels of smoke exposure, wine samples ranged from bound PD concentrations of 0-400 ppb (Figure 2). Grapes from each of the vineyards were classified into five groups of smoke exposure, with only one vineyard showing unsmoked levels (<6 ppb) and four vineyards with elevated smoke exposure (301-400 ppb). Unsurprisingly, vineyards in closer proximity to the larger Ranch and River fires showed higher levels of smoke exposure than other vineyards, supporting a correlation between bound PD levels and probable smoke damage from nearby wildfires. Likewise, samples from more distant vineyards to these fires showed lower levels of bound PDs.
Ultimately, Crews et al. detail a novel method for detection of phenol levels in various wine and grape samples to predict overall wine quality. In theory, this information would be valuable for wine harvesters to understand a crop’s quality before putting in the effort to harvest it; however, it is certainly unlikely an entire crop would be discarded even if the chance of smoke taint was high. Nevertheless, understanding the potential effect of wildfire smoke on grape harvests is important for thorough quality control of the final beverage, especially as climate change continues to have an impact on weather patterns and natural disasters.
References
- Kennison, K. R.; Wilkinson, K. L.; Williams, H. G.; Smith, J. H.; Gibberd, M. R. J. Agric. Food Chem. 2007, 55 (26), 10897– 10901, DOI: 10.1021/jf072509k.
- Hayasaka, Y.; Parker, M.; Baldock, G. A.; Pardon, K. H.; Black, C. A.; Jeffery, D. W.; Herderich, M. J. J. Agric. Food Chem. 2013, 61 (1), 25– 33, DOI: 10.1021/jf305025j.
- Kennison, K. R.; Gibberd, M. R.; Pollnitz, A. P.; Wilkinson, K. L. J. Agric. Food Chem. 2008, 56 (16), 7379– 7383, DOI: 10.1021/jf800927e.
- Phillip Crews, Paul Dorenbach, Gabriella Amberchan, Ryan F. Keiffer, Itzel Lizama-Chamu, Travis C. Ruthenburg, Erin P. McCauley, and Glenn McGourty. Journal of Natural Products. 2022, 85 (3), 547-561. DOI: 10.1021/acs.jnatprod.2c00028.

