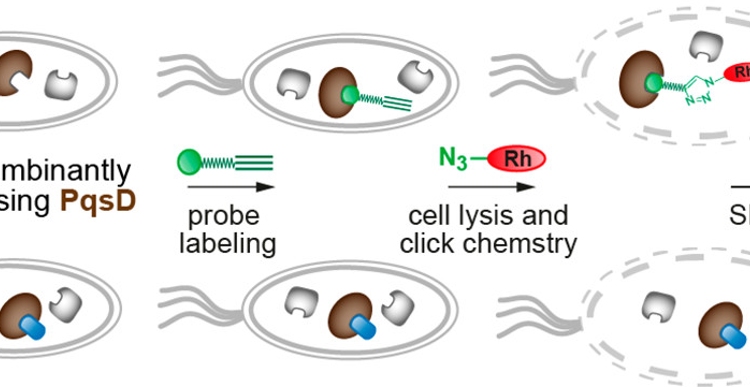
Just as interesting as the detail of how the antibacterial molecules works can be the new methods by which they are discovered. Today’s Chembite is about the development of antibacterial agents in the fight against an infectious bacterium.
Designer Enzymes
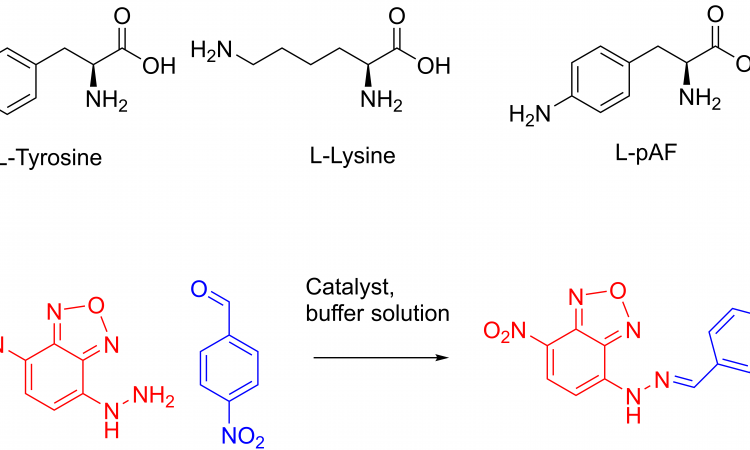
Enzymes are nature’s catalysts. But there are not enough enzymes for all the reactions in the world! How do chemists design enzymes? Let’s take a look!
Can Chemists Cure the Common Cold?
Although getting the common cold is not a nice experience, it is only a relatively minor misfortune for most people. But the consqeuences of a cold can be severe. This paper describes a significant step towards a cure!
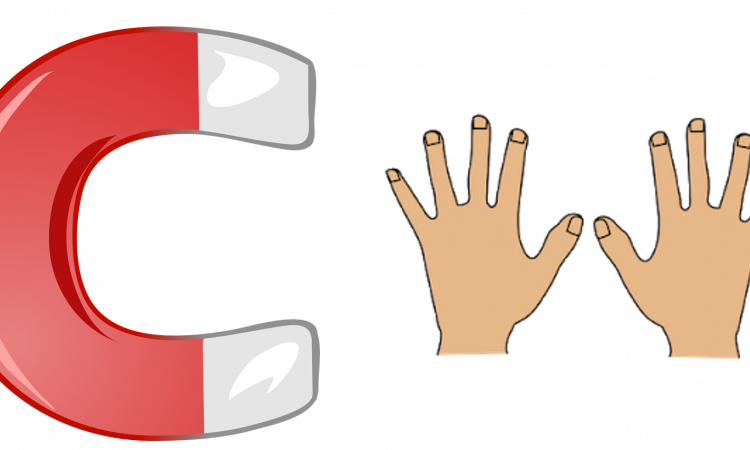
Separating Left- and Right-Handed Molecules With A Magnetic Field
Today’s Chembite looks at a remarkable new way of separating enantiomers – using a magnetic field.
Why Scientists Are Putting Barcodes on Worms
Find out what “photochemical barcodes” are and how they might help us understand complex biological processes.
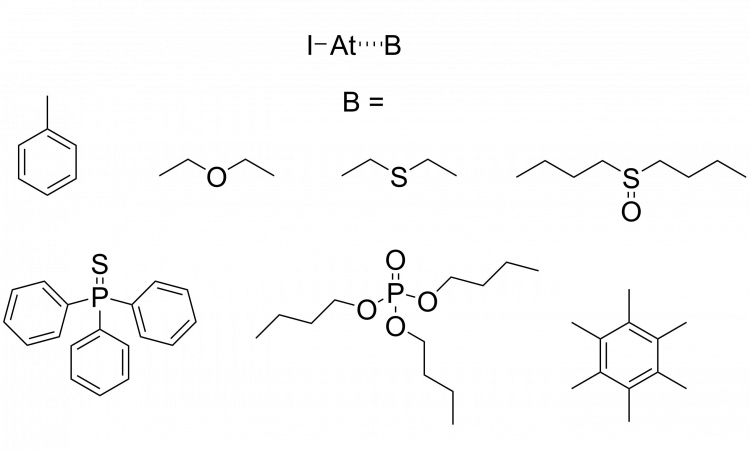
Halogen bonding with astatine: insights into a mysterious element.
How much do you look beyond the top few rows of elements in the periodic table? Prepare to do just that in today’s chembite as we explore some astatine chemistry!
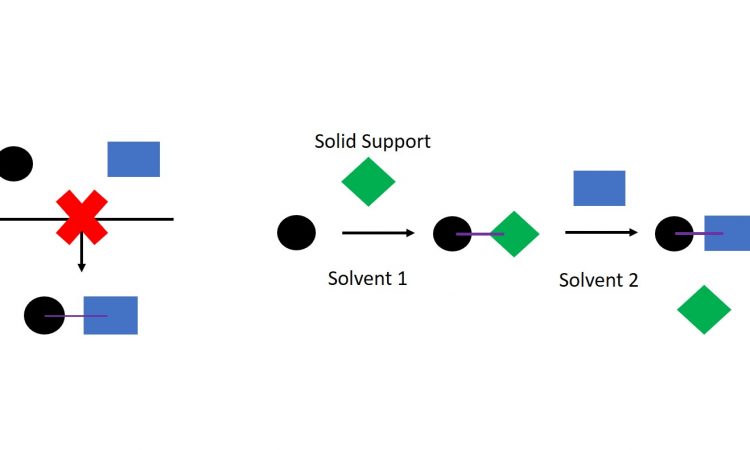
How do you stick hydrophilic and hydrophobic molecules to each other?
We know that complementary functional groups are needed for strong intermolecular interactions, and that thermodynamics favours hydrophilic and hydrophobic groups each keeping themselves to themselves. So, problems can arise when trying to react very hydrophilic and very hydrophobic molecules together. This group of scientists has devised a way around the problem using a technique called solid-phase synthesis.
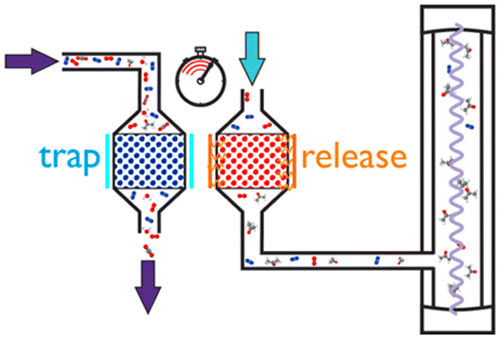
Trap and Release the acetone in your breath: Portable Spectroscopy for Diabetes Diagnosis?
Ask someone what gases they would expect to find in our breath and oxygen, carbon dioxide and water may well be high on the list. But did you realize that acetone could be in there too? A npvel portable device for detecting acetone in breath has been reported.
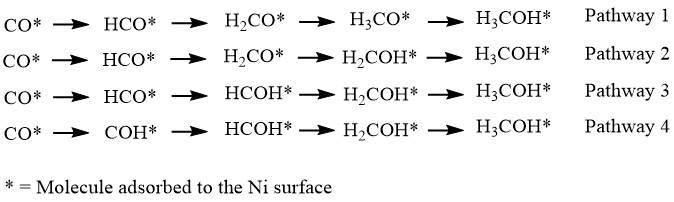
Scratching the Surface: Subsurface Hydrogen Aids CO Hydrogenation to Methanol With Nickel
In today’s Chembite we appreciate and explore some remarkable mechanistic aspects of the hydrogenation of CO at a nickel surface. The paper covered gives the first account of catalytic methanol and formaldehyde production from CO by Ni. But to explain why we need to go deeper than the surface…
The Frustrations of Dihydrogen Activation
Dihydrogen stores a lot of energy. How can we control the release of energy so we can use dihydrogen as a fuel of the future? A little frustration can help.
Carbon Monoxide: Future Therapeutic?
Carbon monoxide is probably one of the last things you would expect to see touted as a new therapeutic. How could this compound be used in medicine?