Article Title: Poly(2,6-anthraquinonyl disulfide) as a high capacity and high-power cathode for rechargeablemagnesium batteries: extra capacity provided bythe disulfide group Authors: Xin Ren, Donggang Tao, Yudi Tang, Yuliang Cao and Fei Xu Journal: Journal of Materials Chemistry A Year: 2023 Image credit: Warut Roonguthai As the…
Going green: Making a vital chemical from nothing but water and sunlight
Hydrogen peroxide can be produced from water using green electrical energy, offering an environmentally friendly route to a vital material.
Clothing Dye or Light Source? Perhaps Both.
Title: From Textile Coloring to Light-emitting Electrochemical Devices: Upcycling of the Isoviolanthrone Vat DyeAuthors: Tatiana Ghanem, Kwang Keat Leong, Hwandong Jang, Alexis Hardouin, Dr. Philippe Blanchard, Prof. Dominik Lungerich, Dr. Pierre Josse, Prof. Eunkyoung Kim, and Dr. Clément CabanetosJournal: Chemistry, An Asian Journal Featured Image: Textile and Fibre Technology, CSIRO…
Unexpected gold nanoparticles arise in ancient buildings through centuries of degradation
New research reveals plausible degradation pathways of metallic gold into nanoparticles through unforeseen corrosion!
From harmful emissions to vital materials: how electrochemistry is making the most of carbon dioxide waste
Carbon dioxide can be converted into useful materials using electrochemistry. This is doubly advantageous, since we can stop carbon dioxide entering the atmosphere and also produce something useful that usually comes from fossil fuels.
A Peek Inside a Fast-Charging Battery
Researchers can now watch what happens to particles inside a battery in real time.
Fungi make molecules for batteries
A active material for redox flow batteries was synthesized from fungi to create a renewable feedstock.
Go Ask Mother Nature—she’s got an answer
An overwhelming majority of scientists are in agreement—and that never happens—something must change before we reach the so-called “point-of-no-return”. The onset of the industrial era (and the associated benefits) encouraged a system that pollutes our environment in search of the largest possible profits. More recently, our voices have gotten louder, and large groups of society have dedicated themselves to uncovering the solutions to these problems. Perhaps, in this regard, Mother Nature still has lessons to offer.
Identifying toxic pesticides with microbial electricity
Some pesticides function similarly to the nerve agent sarin, and their ubiquitous use makes them a constant health hazard if unmonitored. Chemists designed a dual-microbe sensor to selectively and sensitively determine when the hazardous chemicals are nearby.
Cheap Molecule Could Lower the Cost of Batteries
A electroactive anthraquinone has been inexpensively designed with a reasonable but not record breaking stability in redox flow batteries.
Can chemistry overcome incompatibility?
A new way to have incompatible reactions occur in spatially separate regions of a liquid to create methanol from methane.
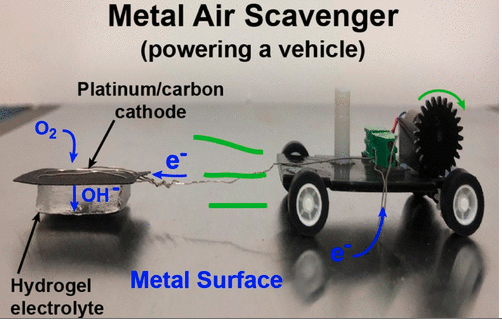
Metal-Air Scavengers Powering Tiny Robots
The smaller the robot, the harder it is to carry a fuel source around. That’s where these metal-air scavengers come in. Powered by oxidizing a metal surface, they could be a useful power source for the tiny robots of the future.
What happens when you add crap to graphene? Literally.
Graphene’s amazing properties make it one of the most popular new materials in recent years. But what if we could improve it with an unlikely additive?
Delicious diagnosis – real-time glucose analysis straight from your gut
Scientists from UCSD and Compultense University developed non-invasive tools to measure gastrointestinal distress, monitoring chemical markers in real-time.
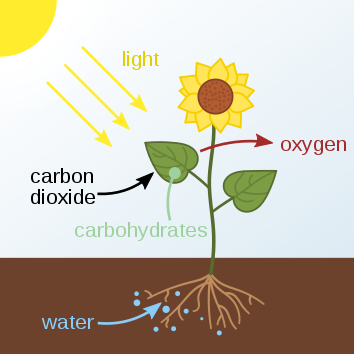
The most promising artificial photosynthesis yet!
Researchers have designed a new way to convert CO2 into fuels that is efficient and cost-effective.
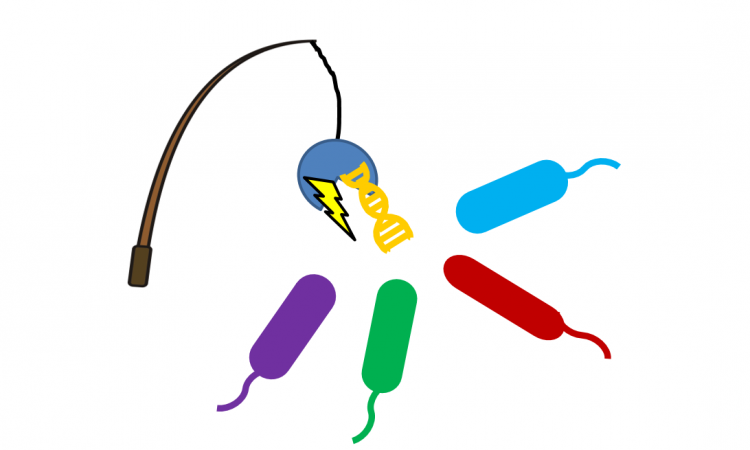
Fishing for bacteria with gold and DNA
What happens when you bring DNA strands, gold nanoparticles, conformation-induced color changes, and a highly-intrusive bacterium together? A field-portable, inexpensive test for the world’s greatest bacterial threats.
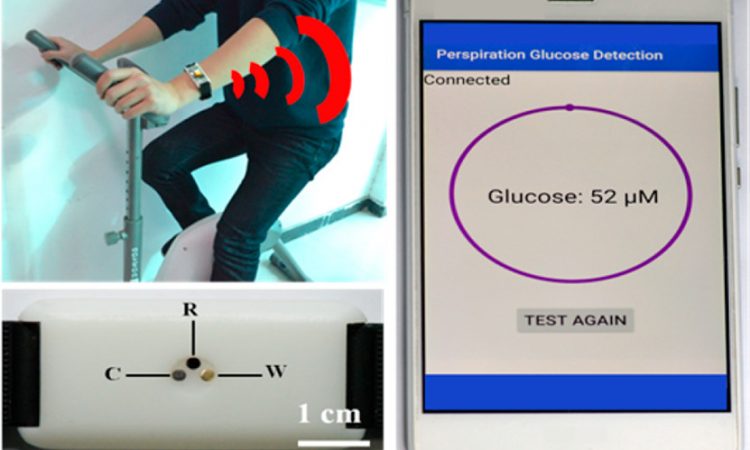
Monitoring Glucose Levels in Sweat with a Wearable Sensor
Measuring blood sugar levels by pricking your finger is painful and inconvenient. Learn about a new wearable device that measures your glucose levels with just your sweat!
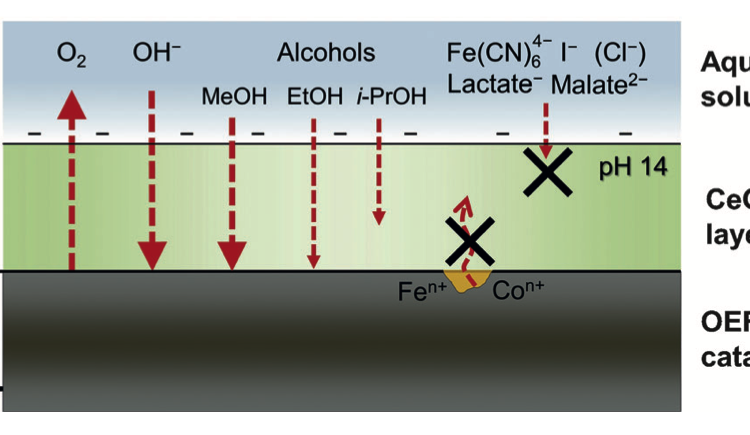
Always Use Protection: Preventing Catalyst Degradation with Coatings
Catalysts are critical components of many industrial processes. Unfortunately, many promising catalysts degrade over time. Here, researchers show that some catalysts can be protected by coating them with another material.
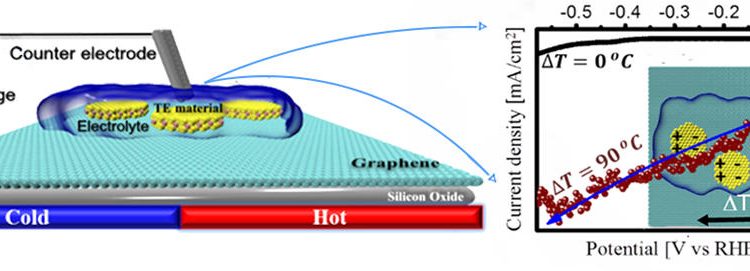
Using Heat to turn Water into Fuel
The development of clean, efficient, and renewable forms of energy is a critical scientific challenge. Plants have already figured out how to do this via photosynthesis. Can we develop a process that mimics this?
Electrochemical Water Oxidation with Cobalt-Based Electrocatalysts from pH 0–14: The Thermodynamic Basis for Catalyst Structure, Stability and Activity
Title: Electrochemical Water Oxidation with Cobalt-Based Electrocatalysts from pH 0–14: The Thermodynamic Basis for Catalyst Structure, Stability and Activity Authors: James B. Gerken , J. Gregory McAlpin , Jamie Y. C. Chen , Matthew L. Rigsby , William H Casey , R. David Britt , and…