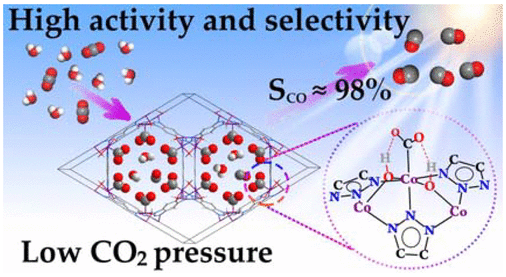
A structure-function relationship has been established for a cobalt containing Metal Organic Framework (MOF) that catalyzes carbon dioxide reduction very efficiently. It has been established that the hydroxyl groups coordinated to the metal co-operates to enhance the catalysis by forming H-bond network with CO2. Let’s learn how the authors performed a systematic and thorough investigation on these MOFs.
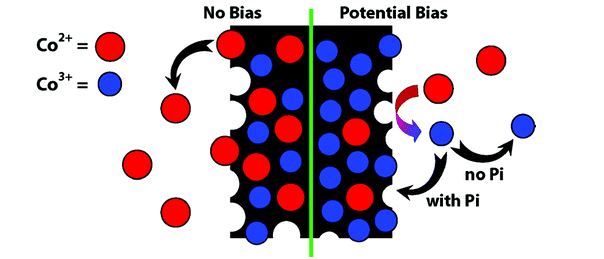
Neutral Water Splitting by Self-Repairing Catalyst
In this work the authors discussed how and why a self-healing mechanism works for their catalyst, as well as how to rationally design other self-healing water oxidation catalysts by better understanding the kinetics of the key catalytic steps.
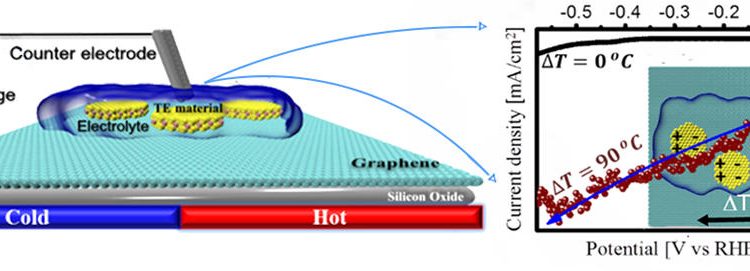
Using Heat to turn Water into Fuel
The development of clean, efficient, and renewable forms of energy is a critical scientific challenge. Plants have already figured out how to do this via photosynthesis. Can we develop a process that mimics this?
Synthetic Biology and Synthetic Chemistry Join Forces
By tailoring mild synthetic chemistry methods to be compatible with living systems, these researchers have made artificial biochemical reactions a reality.
Versatile Surfaces: How to Enhance the Role of Quantum Dots in Photocatalysis
There are lots of ways to use sunlight to achieve sustainable energy goals. Photocatalysts, which can use sunlight to power useful chemical reactions, are of great interest for the production of solar fuels like hydrogen. Read more about how we can use novel nanomaterials as photocatalysts in this Chembite!
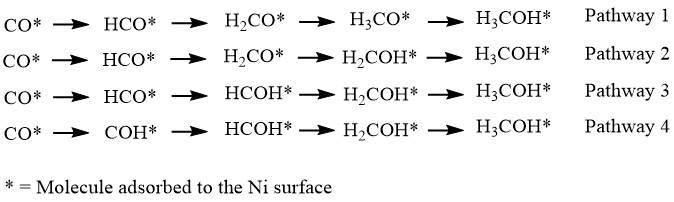
Scratching the Surface: Subsurface Hydrogen Aids CO Hydrogenation to Methanol With Nickel
In today’s Chembite we appreciate and explore some remarkable mechanistic aspects of the hydrogenation of CO at a nickel surface. The paper covered gives the first account of catalytic methanol and formaldehyde production from CO by Ni. But to explain why we need to go deeper than the surface…
Bicarbonate Controlling the Rate of CO2 Reduction
This work shows that bicarbonate (HCO3-) is neither a general acid nor a reaction partner in the rate-limiting step of electrochemical CO2 reduction catalysis mediated by planar polycrystalline Au surfaces. Kinetic modeling studies and electrochemical experiments suggest that it acts as a proton donor in steps past the rate-limiting one and a buffer in the solution.
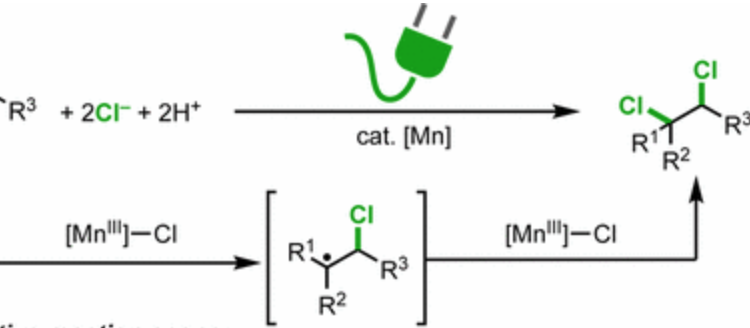
A New Dichlorination of Alkenes: A Radical Twist to a Long-sought Transformation
Title: Electrocatalytic Radical Dichlorination of Alkenes with Nucleophilic Chlorine Sources Authors: Niankai Fu, Gregory S. Sauer, and Song Lin Journal: Journal of the American Chemical Society http://pubs.acs.org/doi/10.1021/jacs.7b09388 Year: 2017 The unsaturated bond of an alkene is one of the most exploited functionalities inside the organic chemist’s…