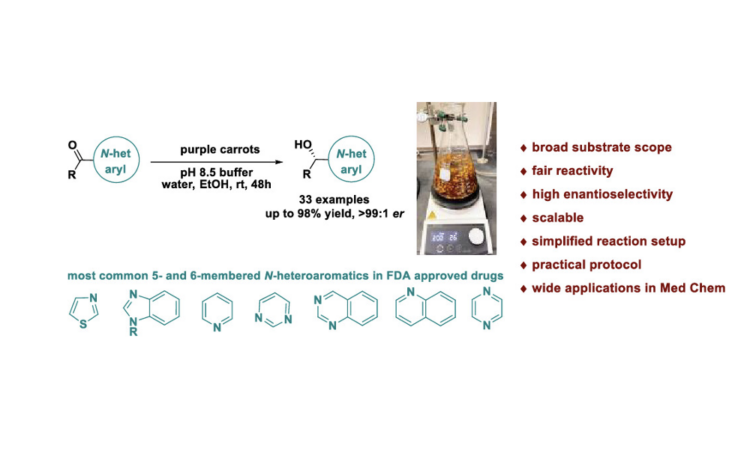
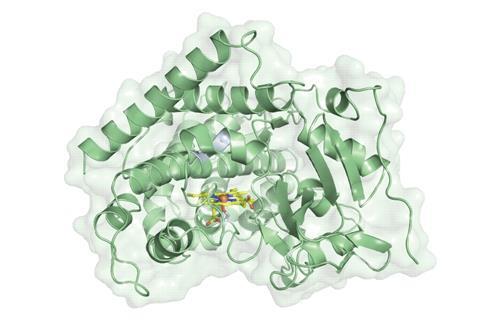
Got carrots? Researchers have developed a new way to enantioselectively reduce ketones to alcohols.
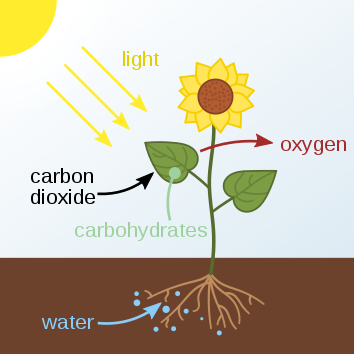
Going green: Making a vital chemical from nothing but water and sunlight
Hydrogen peroxide can be produced from water using green electrical energy, offering an environmentally friendly route to a vital material.
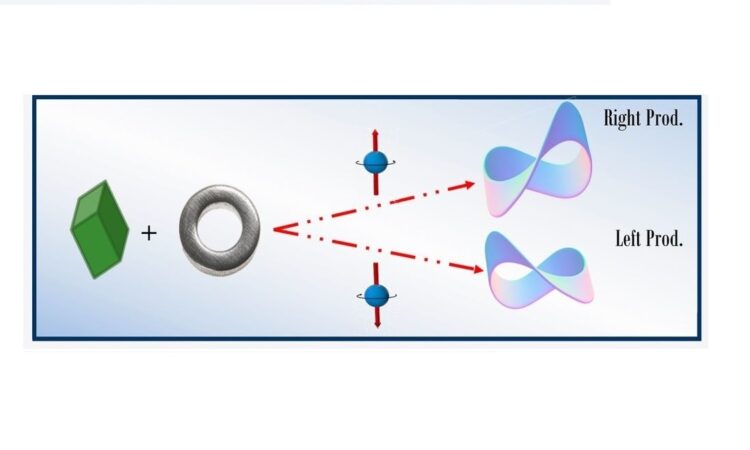
Electron spin selectivity effects can be harvested in asymmetric catalysis
A deeper understanding of spintronics enables the generation of new technologies using chiral molecules.
A Cheap Fix for Diffuse Methane
Converting methane to carbon dioxide could be a key strategy in tackling climate change.
Go Ask Mother Nature—she’s got an answer
An overwhelming majority of scientists are in agreement—and that never happens—something must change before we reach the so-called “point-of-no-return”. The onset of the industrial era (and the associated benefits) encouraged a system that pollutes our environment in search of the largest possible profits. More recently, our voices have gotten louder, and large groups of society have dedicated themselves to uncovering the solutions to these problems. Perhaps, in this regard, Mother Nature still has lessons to offer.
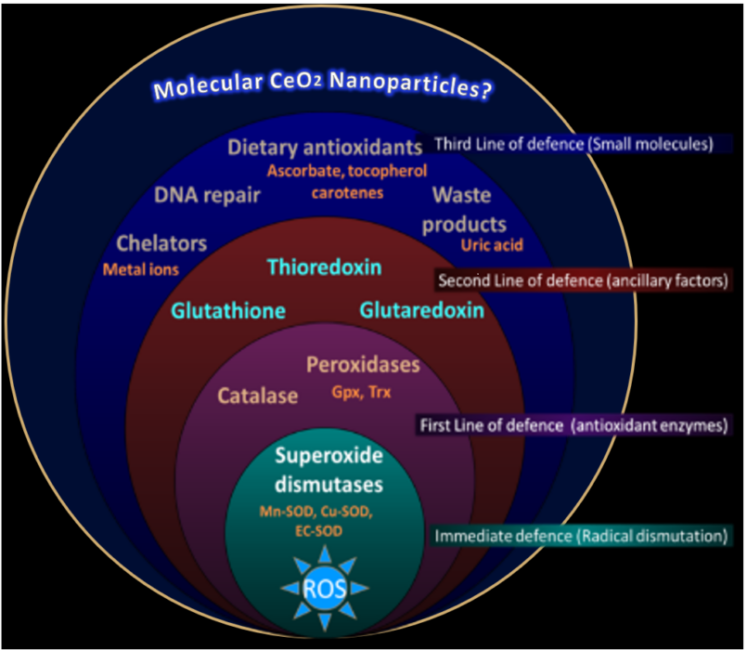
Molecular Nanoparticles: Large Molecules to Prevent Oxidative Stress?
Maybe chemistry can save us—in this case, synthetic chemists have developed a family of lanthanide-oxo molecules and investigate their capabilities as radical species scavengers, with a possible future in the clinic to mitigate various diseases onset by oxidative stress (get antioxidants into your diet, people!).
Polishing a Drug Lead: A Replacement for Opioids Reaches Clinical Trials
Many molecules can decrease enzymatic breakdown of our body’s natural painkillers…but which one is fit to be the best new drug?
Can chemistry overcome incompatibility?
A new way to have incompatible reactions occur in spatially separate regions of a liquid to create methanol from methane.
“Mix n’ Match” Metabolisms
“Jailbreaking” your smartphone is one thing—but what if you could jailbreak nature itself?
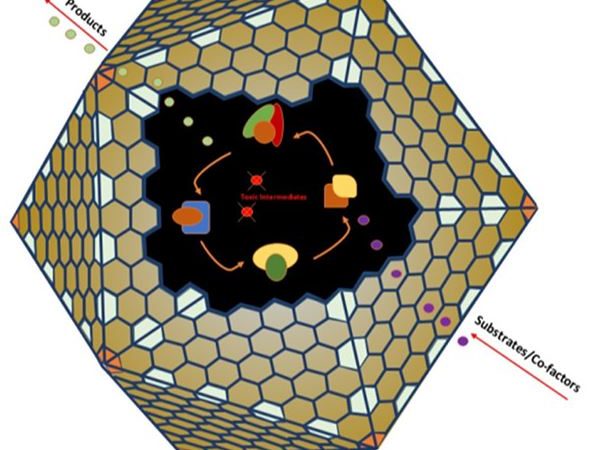
Harnessing an enzyme’s full potential by locking it in a protein cage
Researchers use a naturally crystalline protein to act as a cage to hold another enzyme. This assembly can then be used to turn waste cooking oil into biodiesel.
What happens when you add crap to graphene? Literally.
Graphene’s amazing properties make it one of the most popular new materials in recent years. But what if we could improve it with an unlikely additive?
What’s blacker than black?
There’s a new record holder for the world’s blackest material. Learn about how randomly oriented carbon nanotubes can be used to create a coating darker than anything else ever made!
Enzymes Get Closer to Traditional Organic Chemistry – The Case of Reductive Amination
A clever, two-part biocatalytic strategy grants access to products of reductive amination that can be troublesome to obtain through more traditional synthetic methods.
A more thorough way to study ammonia production
How can researchers eliminate false positives from their research on energy efficient production of ammonia?
Did life come from the depths of the ocean?
Amino acids were found in the Atlantis Massif, under the ocean floor. Is their non-biological synthesis the origin of life?
From Sunscreen to Solar Cells: What’s Happening on TiO2‘s Surface?
The controversy over TiO2’s hydrophilic/hydrophobic transition has been examined in new detail – with researchers concluding that atmospheric molecules can attach onto TiO2’s surface, changing its chemical properties.
A New Duo in Catalysis: Combining Gold Nanoparticles with Enzyme Compartments
Biological catalysts and inorganic catalysts each have their own advantages and it is sometimes difficult to choose one or the other. So why not combine them into a powerful hybrid catalyst? That’s exactly what the researchers did in this recent article from ACS Catalysis.
5 Facts About Catalysts You Might Not Know
Catalysis is a common chemistry concept, but there are many intricacies involved. Check out some of these lesser-known facts!
Evolving Proteins to Make Tiny Carbon Rings
Learn how researchers at Caltech artificially evolved proteins to synthesize some of the most challenging tiny molecules in organic chemistry!
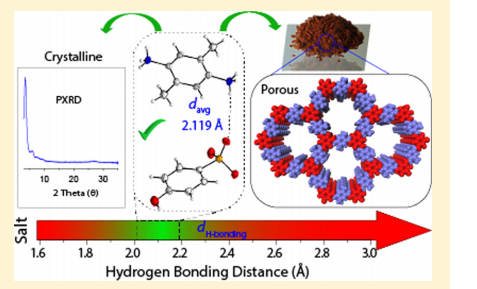
Controlling Crystallinity and Porosity of Covalent Organic Frameworks with Hydrogen Bonding
This work reports a solvent-free solid-to-solid synthesis method for covalent organic frameworks (COFs), which is very promising from a greener and cleaner chemistry standpoint. The researchers found that hydrogen bonding within the starting material plays a key role on the porosity and crystallinity of the final COF.
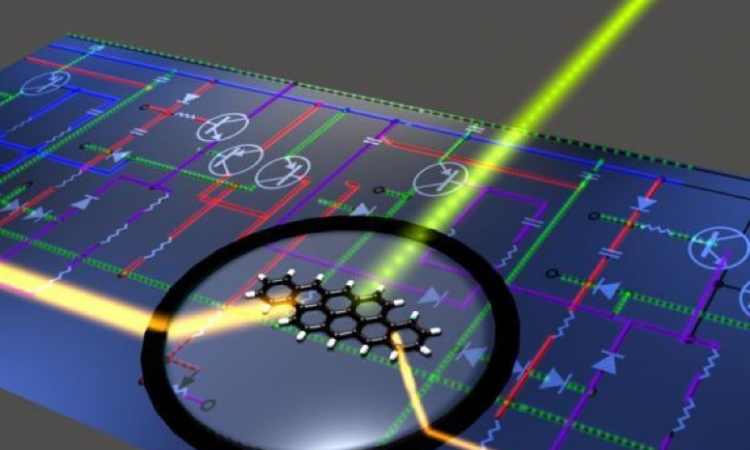
Making computers from molecules – Molecular Turing Machines
In this article, explore the tiny molecules that could be used for computational work in future of smartphones! Inspired by your own body, machines made from molecules could be the next generation of computers!
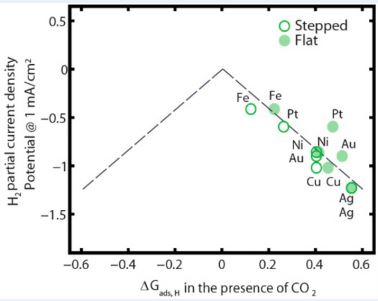
Intricate Balance of Carbon Dioxide and Hydrogen Adsorption for Catalysis
With metal catalysts, we can extract electricity from CO2 – reducing carbon emissions and creating renewable energy tech at the same time! There’s just one little problem, and it’s name is hydrogen…
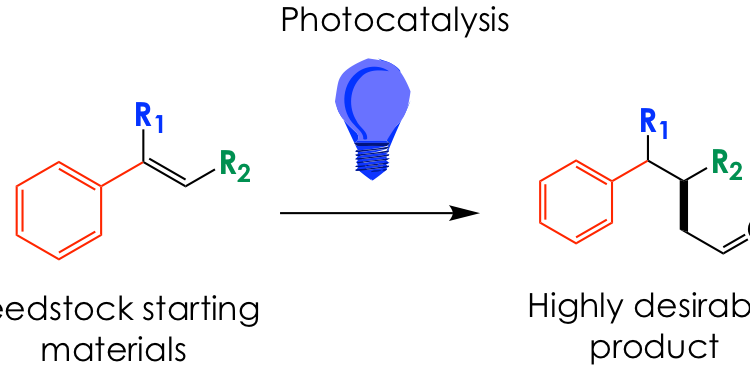
Shining Light on Aldehyde Synthesis
Photoredox catalysis is at it again! This time it is used to synthesize polysubstituted aldehydes – highly useful building blocks – from readily available styrenes and vinyl ethers.
Hitting the Jackpot – Evolving Artificial Biocatalysts Through Randomization
Random change has been powering life’s evolution for billions of years. Can it also power the evolution of artificial biomolecules?
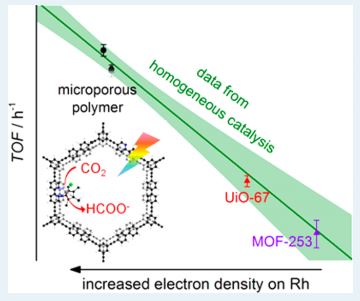
How to Prevent Cannibalism in Catalysts
This work describes an approach to prevent self assimilation of catalysts to increase their lifetime. It also finds a Hammett correlation between different substituents present on the catalysts and the rate of catalysis in both homogeneous and heterogeneous phase.