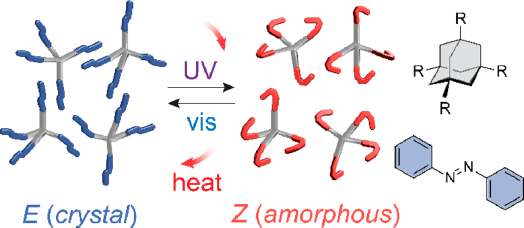
A new class of light-absorbing solids isomerizes to store light energy as heat.
Go Ask Mother Nature—she’s got an answer
An overwhelming majority of scientists are in agreement—and that never happens—something must change before we reach the so-called “point-of-no-return”. The onset of the industrial era (and the associated benefits) encouraged a system that pollutes our environment in search of the largest possible profits. More recently, our voices have gotten louder, and large groups of society have dedicated themselves to uncovering the solutions to these problems. Perhaps, in this regard, Mother Nature still has lessons to offer.
What gives bird feathers such vivid colors?
Birds have a unique way of creating color in their wings and this is how they have become so colorful.
EFFICIENT CHEMISTRY BEHIND THE OLED SCREENS
OLEDs are earth-friendly, energy-efficient, all while being ultra-thin, flexible, and lightweight. They are the future of ultra-efficient lighting. Researchers are now coming up with new techniques to fabricate such sustainable and efficient OLEDs.
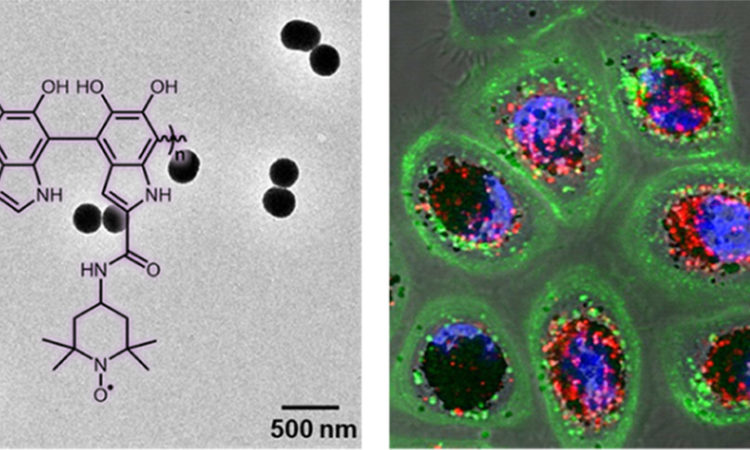
Melanin nanoparticles to better protect our cells
How does melanin protect cells from damaging radiation? How can this ability be improved?
“Mix n’ Match” Metabolisms
“Jailbreaking” your smartphone is one thing—but what if you could jailbreak nature itself?
These artificial muscles don’t mind being “light” weight
Light-responsive substances are all the rage; find out how one research group invented a new class of photomechanical materials.
Make it green, make it bright!
Scientists craft a “greener,” copper-iodide-based ink with amazingly efficient photoluminescent properties
Light me up: can visible light impact forensic luminol reactions?
Crime scene techs use luminol to reveal latent bloodstains – can normal, visible light increase the reaction’s sensitivity?
Is that molecule moving?
Scientists can now observe molecules vibrating
The most promising artificial photosynthesis yet!
Researchers have designed a new way to convert CO2 into fuels that is efficient and cost-effective.
What’s In A Screen? Improving Charge Transfer in Quantum Dot Films
What’s in a screen? It might be tiny particles called quantum dots – a novel material for everything from screens to solar cells. But how do researchers optimize new materials like quantum dot films? One type of spectroscopy – called transient absorption – could help.
From Sunscreen to Solar Cells: What’s Happening on TiO2‘s Surface?
The controversy over TiO2’s hydrophilic/hydrophobic transition has been examined in new detail – with researchers concluding that atmospheric molecules can attach onto TiO2’s surface, changing its chemical properties.
Photocages: Using Light to Deliver Medicine
What will medicine look like 10 years from now? Well, your doctor might be shining a light on you to help target drug delivery in your body. Read more about drug delivery using molecules called photocages inside!
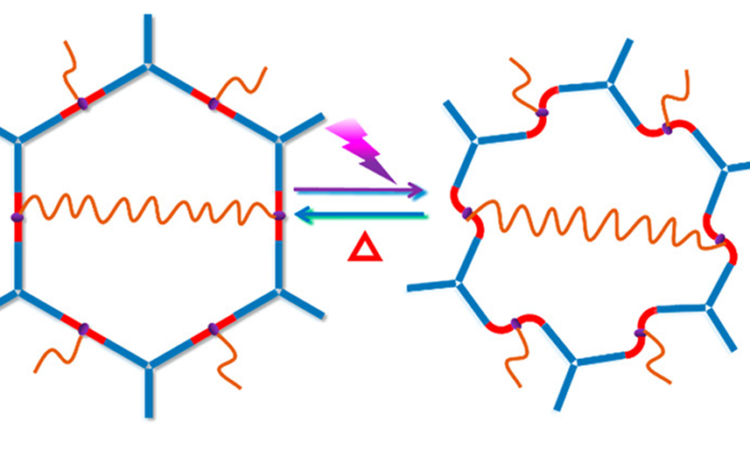
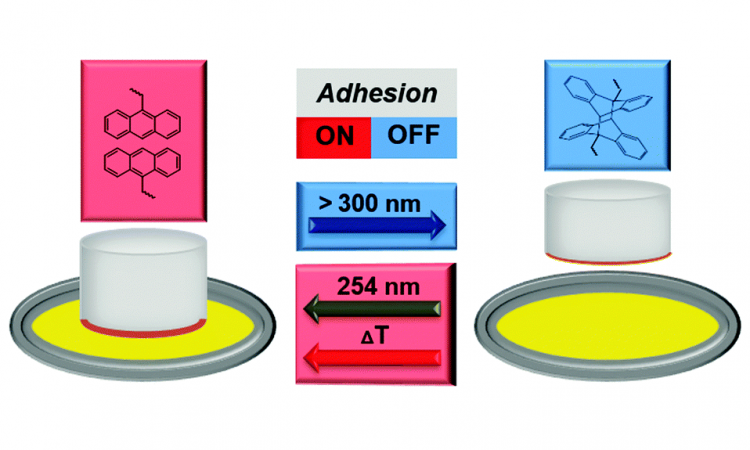
Controlling Adhesives with Light!
Let’s learn about a new material that can be made sticky or not sticky with just light!
Weak Magnetic Fields Affect DNA Repair: A Migratory Bird’s Inner Compass?
Birds and other migratory animals use the Earth’s weak magnetic field to navigate, but what do they use as a compass? While previous research has uncovered some promising candidates, not until now have experiments identified a compass sensitive to fields as weak as Earth’s – DNA repair by photolyase.
Why Scientists Are Putting Barcodes on Worms
Find out what “photochemical barcodes” are and how they might help us understand complex biological processes.
Freedom from Wires: Solar-Charging Perovskite Batteries
Ever had your phone die out of nowhere? Wonder how you’re going to charge your Tesla on your next road trip? Researchers from the University of Cambridge have got your back – they’ve developed a single material that doubles as a battery and a solar cell.
Turn on the lights and get your 3D printouts!
Printing is cool, but 3D printing is cooler! Instead of words on a page, you can print spoons and forks and even houses! And today, you’ll see the coolest 3D printing – printing chains of molecules, simply with light!
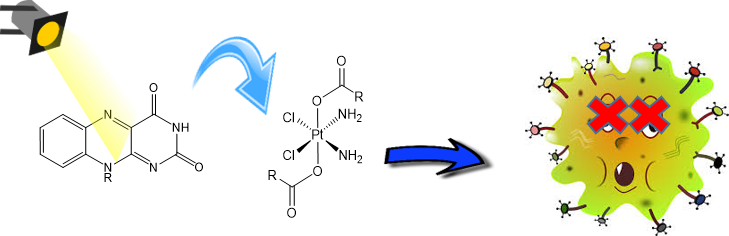
Flavins photoactivation of prodrugs: let’s shine a light
How can flavin and flavoprotein help with cancer therapy? A very nice example of biorthogonal chemistry and its potential.
Household Chemical Products Contribute to Poor Air Quality
Is your deodorant contributing to air pollution and human health risks? Find out here!
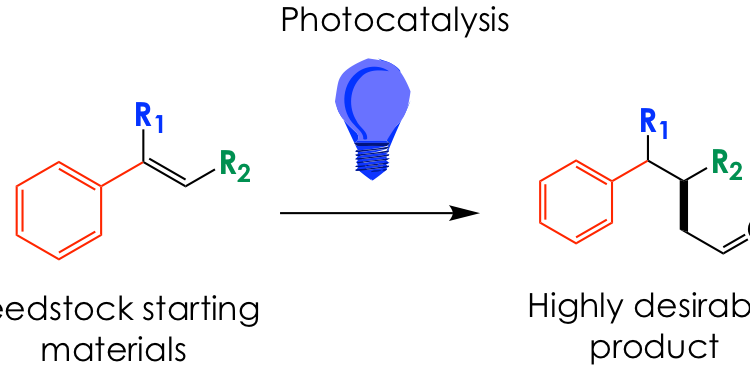
Shining Light on Aldehyde Synthesis
Photoredox catalysis is at it again! This time it is used to synthesize polysubstituted aldehydes – highly useful building blocks – from readily available styrenes and vinyl ethers.
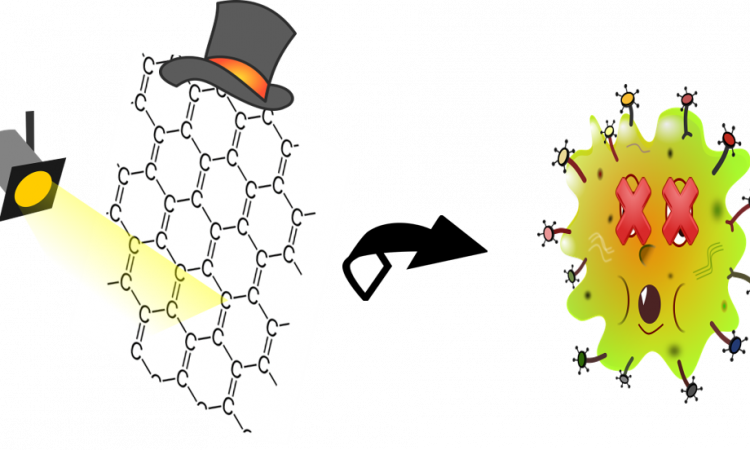
The Thousand Wonders of Graphene: from 2D to 3D Photodrugs!
The authors of this paper can make a photodrug from a special type of graphene.
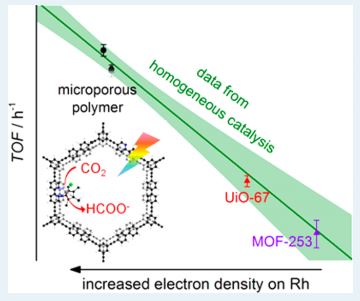
How to Prevent Cannibalism in Catalysts
This work describes an approach to prevent self assimilation of catalysts to increase their lifetime. It also finds a Hammett correlation between different substituents present on the catalysts and the rate of catalysis in both homogeneous and heterogeneous phase.
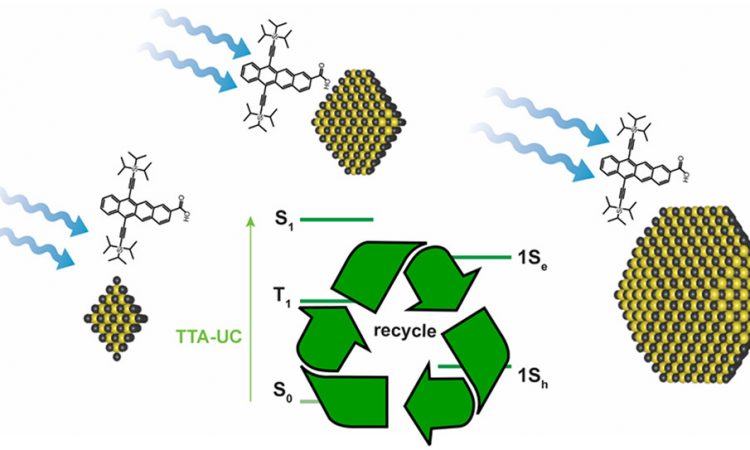
Energy Flow in Quantum Dot – Organic semiconductors
Quantum dots are fascinating super small solids. Highly conjugated tetracene is an electronically active organic molecule. When these two are mixed, electrons bounce around in amazing ways and these researchers found out how.